Buy ARA-290 16mg
$95.00
ARA-290 is variant of erythropoietin which has been shown to decrease inflammatory pathways through paracrine signalling and the innate repair receptors. Research has shown ARA-290 to help reduce HbA1c, improve cholesterol, modulate neuropathic pain and stimulate wound repair.
COA ARA-290 16mg Certificate
ARA-290 16mg: The Advanced Peptide for Neuropathic Pain Relief and Cellular Repair
ARA-290 16mg is a next-generation, research-grade synthetic peptide derived from the protective region of erythropoietin (EPO). Unlike traditional EPO, ARA-290 is specifically engineered to bind the innate repair receptor (IRR) without stimulating erythropoiesis — making it a highly targeted therapeutic candidate for neuropathic pain, inflammation, metabolic dysfunction, and tissue repair. Whether you are a researcher, clinician, or healthcare professional, ARA-290 16mg represents a cutting-edge option in the rapidly evolving field of peptide-based medicine.
HPLC ARA-290 16mg Certificate
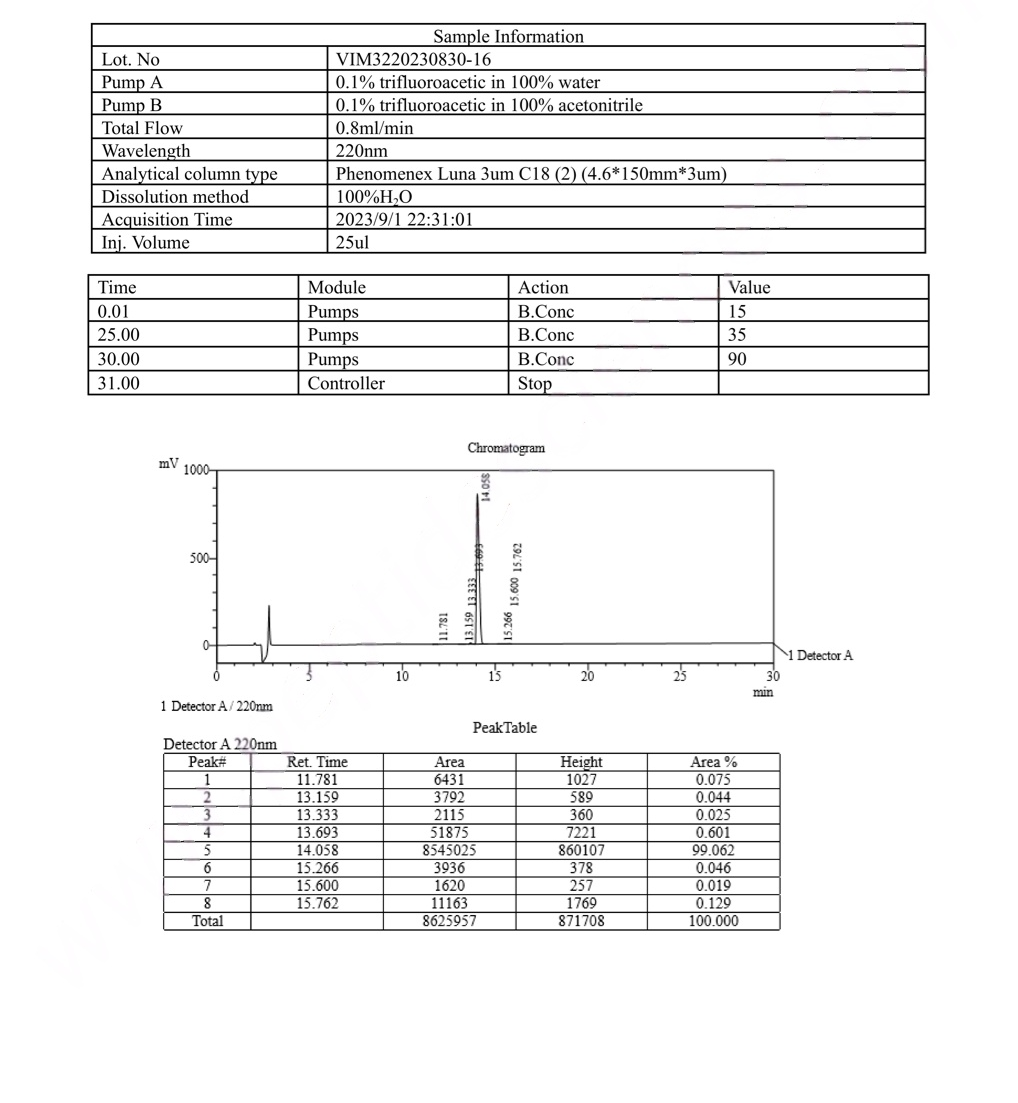
MS ARA-290 16mgCertificate
What Is ARA-290 16mg? Understanding the Innate Repair Receptor Peptide
ARA-290 is an 11-amino-acid cyclic peptide — sometimes called cibinetide — derived from the helical B domain of erythropoietin. Its chemical structure is designed to selectively activate the innate repair receptor (IRR), a heterodimer complex comprising the EPO receptor and the beta-common receptor (βcR). This receptor complex is widely expressed across non-hematopoietic tissues, including peripheral nerves, the pancreas, the heart, and immune cells, which makes ARA-290 a highly versatile compound for systemic cellular protection.
Mechanism of Action: How ARA-290 Works at the Cellular Level
ARA-290 exerts its effects by binding the beta-common receptor (βcR/CD131) component of the IRR. Upon binding, it activates downstream signaling pathways including PI3K/Akt and MAPK/ERK, which mediate anti-apoptotic, anti-inflammatory, and neuro-regenerative responses. Unlike full-length EPO, ARA-290 does not trigger JAK2/STAT5 pathways associated with erythropoiesis, thereby avoiding the risk of polycythemia or thromboembolic events. This selective mechanism profile makes ARA-290 uniquely positioned among repair-oriented peptides.
Key Benefits of ARA-290 16mg: Clinical Research Highlights
Research into ARA-290 has produced a compelling body of evidence across several therapeutic domains. Below are the primary areas where ARA-290 16mg has demonstrated meaningful clinical and preclinical activity:
1. Neuropathic Pain Relief and Nerve Fiber Regeneration
One of the most well-documented effects of ARA-290 is its ability to reduce small fiber neuropathy (SFN) symptoms. In clinical trials involving patients with sarcoidosis-associated SFN, ARA-290 administration led to a statistically
significant reduction in pain scores and improvements in corneal nerve fiber density — a key biomarker of intraepidermal nerve fiber regeneration. These findings suggest ARA-290 may promote peripheral nerve repair and restore sensory function in patients with otherwise treatment-resistant neuropathy.
2. Anti-Inflammatory Activity and Immune Modulation
ARA-290 has demonstrated potent anti-inflammatory properties by suppressing pro-inflammatory cytokines such as TNF-alpha, IL-6, and IL-1beta, while upregulating anti-inflammatory mediators. In macrophage studies, ARA-290 polarizes immune cells toward an M2 (anti-inflammatory) phenotype, which is associated with tissue healing and reduced chronic inflammation. This immunomodulatory capacity makes ARA-290 a candidate for autoimmune and inflammatory disease research.
3. Metabolic Benefits: Blood Sugar Regulation and Pancreatic Support
Emerging research indicates that ARA-290 may play a role in metabolic health, particularly in the context of type 2 diabetes and insulin resistance. The IRR is expressed in pancreatic beta cells, and ARA-290 has been shown to promote beta-cell survival, reduce apoptosis, and potentially enhance insulin secretion under stress conditions. In animal models of type 2 diabetes, ARA-290 treatment was associated with improved glycemic control, pointing toward exciting applications in metabolic medicine.
4. Cardioprotective and Wound Healing Properties
ARA-290 exhibits significant cytoprotective effects in cardiac tissue. Preclinical studies show it reduces infarct size following myocardial ischemia-reperfusion injury by inhibiting cardiomyocyte apoptosis and oxidative stress. Additionally, IRR activation in dermal tissues promotes angiogenesis and accelerates wound closure, suggesting potential applications in wound care and post-surgical recovery. These cardioprotective and regenerative properties further expand ARA-290’s multi-target therapeutic profile.
ARA-290 16mg Dosage, Administration, and Storage
ARA-290 is supplied as a lyophilized (freeze-dried) powder in a 16mg vial, intended for reconstitution with sterile bacteriostatic water before use. Proper handling ensures peptide integrity and experimental reproducibility.
Recommended Research Dosage and Protocols
- Typical dosage used in clinical trials: 4mg administered subcutaneously (SC) once or twice weekly.
- For neuropathy protocols, studies have utilized 4mg SC injections over an 8–28 day period.
- The 16mg vial provides a cost-effective multi-dose supply for extended research protocols.
- Reconstitute with 1–2mL bacteriostatic water; gently swirl — do not shake — to dissolve.
- Refrigerate reconstituted solution at 2–8°C and use within 28 days of reconstitution.
- Always label vials with reconstitution date and concentration for precise dosing
ARA-290 16mg in Peer-Reviewed Research: What the Science Says
ARA-290 has been the subject of multiple peer-reviewed studies and clinical trials, with particular focus on neuropathy, sarcoidosis, and metabolic syndrome. Key publications include work from Leiden University Medical Center (LUMC) in the Netherlands, where double-blind, placebo-controlled trials demonstrated statistically significant improvements in neuropathic pain and quality-of-life metrics in sarcoidosis patients with SFN. These findings were published in journals including Molecular Medicine and PAIN, establishing ARA-290 as a credible clinical-stage peptide with strong translational potential.
Why Choose the 16mg Format for Research Applications?
The 16mg vial size is optimally matched for researchers and clinicians who require multiple doses across extended study periods. Compared to smaller vial formats (2mg or 5mg), the 16mg configuration reduces reconstitution frequency, minimizes compound waste, and offers superior cost-per-dose economics — particularly for longitudinal protocols. Lyophilized powder format ensures maximum stability during transit and long-term dry storage at -20°C, with a typical shelf life of 24 months when properly stored.
ARA-290 16mg Safety Profile and Tolerability
In published clinical trials, ARA-290 has demonstrated a favorable safety and tolerability profile. Unlike full-length recombinant EPO, it does not promote red blood cell production or increase the risk of thromboembolic events. The most frequently reported adverse effects in human studies were mild, transient injection-site reactions. No serious drug-related adverse events were reported in phase I/II trials. Importantly, ARA-290 did not affect hematocrit, hemoglobin, or platelet count, confirming its tissue-protective activity is cleanly separated from erythropoietic effects.
Why Choose Our ARA-290 16mg? Quality, Purity, and Reliability
Our ARA-290 16mg is manufactured to the highest research-grade standards, ensuring consistent purity, accurate dosing, and reproducible results. Every batch is third-party tested using HPLC and mass spectrometry analysis to confirm identity, purity (>98%), and absence of contaminants. We provide full Certificates of Analysis (CoA) with every order, supporting traceability and regulatory compliance for your research documentation.
- Purity: ≥98% verified by HPLC and Mass Spectrometry
- Format: Lyophilized powder in sterile, sealed vial
- Storage: Stable for 24+ months at -20°C (dry); 28 days post-reconstitution at 2–8°C
- Comes with full Certificate of Analysis (CoA) and lot traceability
- Fast, discreet international shipping with cold-chain options available
- Expert customer support for research protocol guidance
Frequently Asked Questions About ARA-290 16mg
Is ARA-290 the same as Cibinetide?
Yes. ARA-290 is the research designation for cibinetide, the INN (International Nonproprietary Name) assigned to this compound. Both names refer to the same 11-amino-acid cyclic peptide derived from the helix-B region of EPO.
Does ARA-290 Stimulate Red Blood Cell Production?
No. ARA-290 was specifically designed to avoid erythropoietic signaling. It does not activate the classical EPO receptor homodimer (EPORh) responsible for red blood cell production, making it hematologically neutral.
What Research Areas Is ARA-290 16mg Best Suited For?
ARA-290 is most actively researched for neuropathic pain (particularly small fiber neuropathy), inflammatory and autoimmune conditions, metabolic disorders including type 2 diabetes, cardioprotection, and wound healing. The 16mg format supports sustained multi-dose protocols across all these domains.
How Should ARA-290 16mg Be Stored Before Reconstitution?
Store the lyophilized powder at -20°C in a dark, dry environment. Avoid freeze-thaw cycles. Once reconstituted, store at 2–8°C and use within 28 days. Do not freeze reconstituted solution.
Order ARA-290 16mg Today — Premium Research-Grade Peptide
Advance your research with confidence. ARA-290 16mg is in stock, batch-tested, and ready to ship. Add to your cart now and receive your Certificate of Analysis with every order. For bulk orders, institutional pricing, or custom research inquiries, contact our specialist team. We are committed to supporting your scientific mission with the highest-quality peptides on the market.
4 reviews for Buy ARA-290 16mg
Add a review Cancel reply
Related products
Peptides
Peptides
Peptides
Peptides
Peptides
Peptides


















Thomas Reed –
Used for nerve pain study—very promising results in reducing discomfort. Highly recommend for neuropathy support.
Used for nerve pain study—very promising results in reducing discomfort. Highly recommend for neuropathy support. –
osanders.peptidelab@outlook.co
Kelly Ross –
Really helped manage post-injury nerve sensitivity. Subtle but impactful
Daniel Phillips –
A clean product that shows consistent quality. Results matched expectations