Cagrilintide 10mg
$119.00
Cagrilintide 10mg — a long-acting amylin analog for research into appetite suppression, weight loss, metabolic health, and next-generation obesity management.
Cagrilintide 10mg: The Next-Generation Long-Acting Amylin Analog Advancing Obesity and Metabolic Research
The science of weight regulation is undergoing a profound transformation. Where earlier generations of research focused primarily on caloric restriction and behavioral modification, today’s most compelling breakthroughs are emerging from the biology of satiety hormones — the peptides and their analogs that communicate directly with the brain to regulate hunger, food intake, gastric emptying, and energy balance at the deepest physiological level. At the forefront of this scientific revolution stands Cagrilintide, a potent, ultra-long-acting amylin analog that is redefining what is possible in obesity and metabolic disease research.
Cagrilintide 10mg is a research-grade peptide formulated for scientific investigation into the amylin receptor pathway and its role in appetite regulation, body weight management, glycemic control, and cardiometabolic health. Developed by Novo Nordisk as part of an ambitious dual-hormone research program, Cagrilintide — also known by its investigational designation AM833 — has advanced through multiple phases of clinical study, demonstrating impressive efficacy as a standalone agent and remarkable synergistic potential when combined with GLP-1 receptor agonists such as semaglutide. This product is intended exclusively for qualified research purposes.
What Is Cagrilintide? Understanding the Amylin Analog Mechanism
Cagrilintide is a synthetic, fatty-acid-acylated analog of human amylin — a 37-amino-acid peptide co-secreted with insulin by pancreatic beta cells in response to food intake. In its natural form, amylin plays a critical and often underappreciated role in postprandial glucose regulation and satiety signaling. It slows gastric emptying, suppresses glucagon secretion during meals, and acts on amylin receptors in the area postrema and nucleus tractus solitarius of the brainstem to reduce food intake and promote feelings of fullness.
Despite its physiological importance, native human amylin is poorly suited to therapeutic or research use due to its tendency to aggregate into amyloid fibrils — the same type of protein misfolding implicated in Alzheimer’s disease and type 2 diabetes progression. The first clinically approved amylin analog, pramlintide, addressed this aggregation problem but required multiple daily injections due to its very short half-life. Cagrilintide represents a major technological leap forward: through selective amino acid substitutions and the attachment of a C18 fatty diacid moiety, it achieves dramatically enhanced receptor binding affinity, structural stability, and a half-life of approximately seven days — enabling once-weekly subcutaneous dosing with sustained receptor engagement throughout the dosing interval.
The Amylin Receptor System: Why It Matters for Obesity Research
The amylin receptor is a complex of the calcitonin receptor combined with receptor activity-modifying proteins (RAMPs), most importantly RAMP1 and RAMP3. This receptor system is expressed throughout the central nervous system, particularly in regions involved in energy homeostasis including the hypothalamus, brainstem, and reward pathways. Activation of the amylin receptor system produces a constellation of metabolic effects that are highly relevant to obesity research: reduced meal size and frequency, decreased caloric intake, suppressed glucagon secretion, slowed gastric emptying, improved postprandial glucose control, and reduced body weight through both reduced energy intake and potentially increased energy expenditure.
What makes the amylin pathway particularly exciting from a research standpoint is its complementarity with other satiety hormone pathways. The GLP-1 receptor system — the target of semaglutide, liraglutide, and other currently approved obesity medications — and the amylin receptor system work through largely non-overlapping central mechanisms, suggesting that dual activation could produce additive or even synergistic effects on food intake and body weight beyond what either pathway can achieve alone. This hypothesis has driven the development of the investigational combination CagriSema, pairing Cagrilintide with semaglutide, which has shown particularly compelling results in early phase clinical research.
Structural Features That Enable Once-Weekly Dosing
The extended half-life and receptor affinity of Cagrilintide are the direct result of deliberate structural engineering. The native amylin sequence was modified through substitution of prolines at positions 25, 28, and 29 to prevent beta-sheet aggregation. Additional amino acid modifications enhance receptor selectivity and binding stability. The attachment of a C18 fatty diacid chain via a linker enables reversible binding to albumin in the bloodstream, dramatically extending the peptide’s circulating half-life from minutes — as in native amylin — to approximately seven days. This albumin binding mechanism is similar to the approach used in the development of semaglutide and insulin degludec, both of which have demonstrated that fatty acid acylation can transform short-acting peptides into clinically practical long-acting agents.
Key Research Findings: What Clinical Studies Reveal About Cagrilintide
Cagrilintide has been evaluated in multiple clinical trials sponsored by Novo Nordisk, generating a substantial body of evidence regarding its pharmacokinetics, pharmacodynamics, safety profile, and weight loss efficacy. The following summarizes the most significant findings from the peer-reviewed and publicly disclosed research to date.
Phase 1 Pharmacokinetic and Safety Studies
Initial Phase 1 studies in healthy volunteers and individuals with obesity characterized the pharmacokinetic profile of Cagrilintide across a range of doses from 0.3mg to 4.5mg administered subcutaneously. These studies confirmed the predicted once-weekly dosing profile, with peak plasma concentrations reached within 24 hours of injection and a half-life consistent with approximately seven-day dosing intervals. The peptide demonstrated dose-proportional exposure and linear pharmacokinetics across the studied dose range. The safety profile in Phase 1 was consistent with the known effects of amylin receptor activation — primarily gastrointestinal effects including nausea and vomiting, particularly during dose escalation — with no serious adverse events attributable to the drug identified at doses up to 4.5mg.
Phase 2 Efficacy Studies in Obesity
A randomized, double-blind, placebo-controlled Phase 2 trial published in The Lancet evaluated Cagrilintide at doses of 0.3mg, 0.6mg, 1.2mg, 2.4mg, and 4.5mg administered once weekly for 26 weeks in adults with overweight or obesity but without type 2 diabetes. The primary endpoint was percent change in body weight from baseline. Dose-dependent weight loss was observed across all active treatment groups, with the highest dose group (4.5mg) achieving a mean weight loss of approximately 10.8 percent from baseline — a clinically meaningful result comparable to established pharmacotherapies and achieved in just six months of treatment. Reductions in waist circumference, blood pressure, and fasting triglycerides were also observed, suggesting broader cardiometabolic benefits beyond weight reduction alone.
The CagriSema Combination: Synergistic Dual Hormone Research
Perhaps the most scientifically significant research involving Cagrilintide has explored its combination with semaglutide — the GLP-1 receptor agonist that has transformed obesity pharmacotherapy. The investigational agent CagriSema, combining Cagrilintide and semaglutide 2.4mg in a single once-weekly injection, has demonstrated weight loss outcomes in Phase 2 research that substantially exceed those achieved by either agent alone. In a Phase 2 trial, the CagriSema combination produced weight reductions of approximately 15 to 17 percent over 32 weeks — results that point toward the potential for dual amylin and GLP-1 receptor co-activation to define the next generation of pharmacological weight management research. Phase 3 trials of CagriSema under the name REDEFINE are ongoing.
Effects on Glycemic Control and Cardiometabolic Markers
Beyond its weight loss effects, Cagrilintide has demonstrated favorable impacts on multiple cardiometabolic markers in research settings. Reductions in HbA1c, fasting plasma glucose, and postprandial glucose excursions have been documented, consistent with the known mechanism of amylin receptor activation including suppression of postprandial glucagon and slowed gastric emptying. Improvements in lipid profiles — particularly reductions in triglycerides — and modest reductions in blood pressure have also been observed, suggesting a comprehensive cardiometabolic benefit profile that extends well beyond simple weight reduction.
Mechanism of Action: How Cagrilintide Regulates Appetite and Body Weight
Understanding the precise mechanisms through which Cagrilintide produces its metabolic effects is essential for researchers working in the fields of obesity biology, neuroendocrinology, and metabolic pharmacology. The following outlines the primary and secondary mechanisms relevant to its research applications.
Central Appetite Suppression via Brainstem and Hypothalamic Circuits
The primary anorectic action of Cagrilintide is mediated through amylin receptor activation in the area postrema — a circumventricular organ in the brainstem that lacks a blood-brain barrier and therefore has direct access to circulating peptides. Amylin receptor activation in this region triggers downstream signaling through the nucleus tractus solitarius and subsequently influences hypothalamic feeding circuits including the arcuate nucleus, where it interacts with both orexigenic (NPY/AgRP) and anorexigenic (POMC/CART) neuronal populations. The net effect is a reduction in meal size, meal frequency, and total daily caloric intake that is sustained throughout the dosing interval due to Cagrilintide’s extended half-life.
Gastric Emptying Modulation
Amylin receptor activation slows the rate of gastric emptying — the pace at which food moves from the stomach into the small intestine. This effect prolongs postprandial satiety, blunts postprandial glucose excursions by spreading glucose absorption over a longer time period, and reduces the rate at which food-derived signals terminate the feeling of fullness. In research settings, this gastric emptying effect of Cagrilintide is measurable by gastric scintigraphy and contributes meaningfully to its overall impact on caloric intake and glycemic control.
Glucagon Suppression and Postprandial Glucose Regulation
Native amylin suppresses glucagon secretion from pancreatic alpha cells during the postprandial period — an effect that is inappropriately absent in type 2 diabetes, contributing to excessive hepatic glucose output and postprandial hyperglycemia. Cagrilintide recapitulates this effect with its extended duration of action, providing sustained glucagon suppression that contributes to improved glycemic control across the full once-weekly dosing interval. This mechanism is complementary to but distinct from GLP-1 receptor agonism, helping explain the additive glycemic benefits observed when Cagrilintide is combined with semaglutide in the CagriSema research program.
Research Applications of Cagrilintide 10mg
Cagrilintide 10mg is intended for use by qualified scientific researchers investigating the following areas of metabolic and neuroendocrine biology:
- Amylin receptor pharmacology — characterizing receptor binding, downstream signaling cascades, and tissue distribution of amylin receptor subtypes
- Obesity and energy homeostasis research — studying the central and peripheral mechanisms of amylin-mediated appetite suppression and body weight regulation
- Combination hormone therapy research — investigating synergistic interactions between amylin receptor agonism and GLP-1, GIP, or other metabolic peptide pathways
- Glycemic control and postprandial metabolism — exploring the role of amylin receptor activation in glucagon suppression and glucose homeostasis
- Cardiometabolic disease research — examining the effects of sustained amylin receptor activation on cardiovascular risk factors including lipids, blood pressure, and inflammatory markers
- Pharmacokinetic and pharmacodynamic modeling — studying the behavior of fatty-acid-acylated peptides and albumin-binding half-life extension strategies
- Peptide formulation science — investigating stabilization, aggregation prevention, and delivery system optimization for long-acting amylin analogs
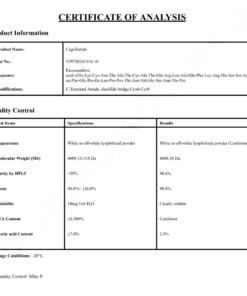
Product Specifications and Quality Standards
Our Cagrilintide 10mg research product is manufactured to the highest standards of purity and quality consistent with research-grade peptide production. All specifications are verified through independent analytical testing prior to release.
Analytical Specifications
- Peptide content: Cagrilintide (AM833) — minimum 98% purity by HPLC analysis
- Molecular formula and mass confirmed by high-resolution mass spectrometry
- Sterility tested — endotoxin levels below 1 EU/mg by LAL assay
- Water content determined by Karl Fischer titration
- Certificate of Analysis provided with every batch confirming all analytical parameters
Storage and Handling
- Store lyophilized peptide at -20 degrees Celsius protected from light and moisture
- Reconstitute with sterile bacteriostatic water or sterile saline for research use
- Use reconstituted solution within 28 days when stored at 2 to 8 degrees Celsius
- Avoid repeated freeze-thaw cycles which may compromise peptide integrity
- Handle under aseptic conditions appropriate for research-grade biological materials
Regulatory and Research Use Statement
Cagrilintide 10mg is supplied exclusively for in vitro and in vivo scientific research purposes. It is not approved for human therapeutic use, veterinary use, or any application outside qualified research settings. Researchers are responsible for compliance with all applicable institutional, national, and international regulations governing the acquisition, storage, handling, and use of research-grade peptides. This product is not intended for self-administration.
Advance Your Metabolic Research With Cagrilintide 10mg
The amylin receptor pathway represents one of the most promising and scientifically validated frontiers in obesity and metabolic disease research. Cagrilintide 10mg places this frontier within reach — providing qualified researchers with a rigorously characterized, high-purity, long-acting amylin analog that mirrors the investigational compound at the center of some of the most exciting clinical development programs in modern endocrinology.
Whether your research focus is receptor pharmacology, energy homeostasis, combination hormone signaling, or cardiometabolic biology, Cagrilintide 10mg delivers the structural integrity, analytical documentation, and research-grade quality your work demands. Order today and contribute to the next chapter of obesity and metabolic science.
Be the first to review “Cagrilintide 10mg” Cancel reply
Related products
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide
Popular products
Peptide














Reviews
There are no reviews yet.