Selank spray 10 mg
$90.00
Selank Nasal Spray 10mg — rapid-onset intranasal nootropic peptide for anxiety relief, focus, memory, and stress resilience. Lab-tested. Discreet worldwide shipping.
Selank Nasal Spray 10mg — Rapid-Onset Intranasal Nootropic Peptide for Anxiety Relief, Cognitive Enhancement, and Stress Resilience
Selank Nasal Spray 10mg delivers the clinically validated Russian nootropic heptapeptide Selank through a precision intranasal spray device — the same route of administration used in the approved pharmaceutical formulation in Russia and Ukraine. By bypassing the gastrointestinal tract entirely and utilising the direct olfactory and trigeminal nerve pathways from the nasal mucosa to the brain, intranasal Selank achieves faster onset, higher central bioavailability, and more efficient brain delivery than any oral administration method. Each spray bottle contains 10mg of pharmaceutical-grade Selank (Thr-Lys-Pro-Arg-Pro-Gly-Pro) dissolved in a sterile, isotonic nasal solution, providing a convenient, needle-free, and scientifically grounded delivery format for research into this exceptional anxiolytic and cognitive-enhancing neuropeptide.
What Is Selank Nasal Spray 10mg?
Selank is a synthetic heptapeptide with the amino acid sequence Thr-Lys-Pro-Arg-Pro-Gly-Pro, developed by the Institute of Molecular Genetics of the Russian Academy of Sciences as a next-generation anxiolytic agent that combines powerful anti-anxiety properties with simultaneous cognitive enhancement — a combination that conventional anxiolytics like benzodiazepines are fundamentally incapable of providing. Based on the endogenous immunopeptide tuftsin and extended with a stabilising Pro-Gly-Pro C-terminal sequence for improved metabolic resistance, Selank has accumulated over two decades of clinical use in Russia and Ukraine as an approved pharmaceutical drug for generalised anxiety disorder (GAD), asthenia, and phobic disorders under the brand name Selank.
The nasal spray formulation of Selank represents the clinically approved and gold-standard delivery route for this peptide. When Selank is administered intranasally, it is absorbed through the highly vascularised nasal mucosa and transported to the central nervous system via two distinct pathways: the olfactory nerve route, which provides direct axonal transport from the olfactory epithelium to the olfactory bulb and limbic system; and the trigeminal nerve route, which delivers peptide directly to the brainstem and cerebellum. These neuroanatomical pathways bypass the blood-brain barrier entirely, delivering Selank directly to the brain structures most relevant to its anxiolytic and nootropic effects with minimal systemic exposure and maximum central nervous system bioavailability.
Each 10mg spray bottle is formulated to deliver a consistent, precisely metered dose with every actuation, providing reliable and reproducible administration for structured research protocols.
Why Intranasal Delivery Is the Optimal Route for Selank
The Blood-Brain Barrier Challenge for Peptides
One of the fundamental pharmacokinetic challenges facing all peptide-based neuroactive compounds is the blood-brain barrier (BBB) — the highly selective cellular membrane that separates circulating blood from the brain’s extracellular fluid and tightly restricts the passage of large, hydrophilic molecules including peptides. Most peptides administered systemically are unable to cross the BBB in therapeutically meaningful quantities, limiting their central nervous system bioavailability and requiring higher systemic doses to achieve adequate brain concentrations. This BBB limitation is particularly relevant for heptapeptides like Selank, which are too large and too polar for efficient passive transcellular BBB crossing.
Direct Nose-to-Brain Transport — The Olfactory Pathway
Intranasal delivery elegantly circumvents the BBB through the olfactory pathway. The olfactory epithelium in the upper nasal cavity is the only location in the human body where neurons — the olfactory receptor neurons — are directly exposed to the external environment with their axons projecting uninterrupted into the brain. Peptides absorbed across the olfactory epithelium are taken up by olfactory receptor neuron axons and transported via axonal flow directly into the olfactory bulb, from where they distribute into the limbic system, hippocampus, amygdala, and prefrontal cortex — the precise brain structures that mediate Selank’s anxiolytic, memory-enhancing, and mood-stabilising effects. This direct neuroanatomical highway from nose to brain is the mechanism underlying intranasal Selank’s rapid onset and high central bioavailability.
Trigeminal Nerve Pathway — Additional Brain Delivery
Complementing the olfactory pathway, the trigeminal nerve — the fifth cranial nerve — innervates the nasal mucosa extensively and provides an additional direct transport route from nasal mucosa to brain. Trigeminal nerve-mediated transport delivers compounds to the brainstem and cerebellum, broadening the brain distribution of intranasally administered peptides beyond the olfactory-bulb-to-limbic-system axis. Together, the olfactory and trigeminal pathways provide Selank Nasal Spray with comprehensive brain-wide distribution through direct neuronal transport, achieving central bioavailability and speed of onset that neither subcutaneous injection nor any oral administration can match for this specific peptide.
Onset Speed Advantage Over Subcutaneous Injection
While subcutaneous injection of Selank achieves systemic absorption within minutes, intranasal delivery produces detectable central effects even faster due to the direct nose-to-brain neuronal transport pathway that bypasses both systemic circulation and the BBB. Published research on intranasal peptide pharmacokinetics consistently demonstrates that olfactory-route delivery produces earlier peak central concentrations than equivalent subcutaneous doses. For Selank specifically — whose approved pharmaceutical formulation is an intranasal spray — this rapid central delivery is a design feature rather than a secondary benefit, making the nasal spray format the pharmacokinetically optimal delivery system for this compound.
Key Benefits of Selank Nasal Spray 10mg
1. Rapid-Onset Anxiolytic Relief Without Sedation
Selank Nasal Spray delivers its well-documented anxiolytic effects with a speed of onset that makes it highly suitable for acute anxiety management research protocols as well as sustained daily administration studies. Via the intranasal route, anxiolytic effects are typically apparent within 10 to 20 minutes of administration — comparable to or faster than subcutaneous injection — and without any sedation, psychomotor impairment, or cognitive blunting. This non-sedating anxiolytic profile has been confirmed in multiple Russian clinical trials comparing Selank to benzodiazepine comparators, where Selank demonstrated equivalent anxiety reduction with dramatically superior cognitive and functional tolerability.
2. Enhanced Cognitive Performance — Memory, Focus, and Processing Speed
Unlike benzodiazepines, which impair memory consolidation and cognitive processing as a direct consequence of their GABAergic mechanism, Selank simultaneously reduces anxiety and enhances multiple dimensions of cognitive performance. Research with Selank nasal spray has documented improvements in working memory capacity, attention span, information retrieval speed, and learning consolidation — effects mediated through BDNF upregulation, dopaminergic modulation in the prefrontal cortex, and the reduction of anxiety-driven cognitive noise that impairs information processing in stressed individuals. The nasal spray’s rapid central delivery means these cognitive enhancement effects are available within minutes of administration, making it particularly valuable for research into acute cognitive performance under anxious or stressful conditions.
3. Stress Resilience and HPA Axis Modulation
Selank modulates the hypothalamic-pituitary-adrenal (HPA) axis — the neuroendocrine stress response system — blunting excessive cortisol reactivity to psychological stressors while preserving the normal adaptive stress response. This HPA modulation supports long-term stress resilience, prevents the hippocampal neuronal damage associated with chronic cortisol excess, and protects immune function from stress-induced suppression. Via the intranasal route, Selank’s HPA-modulating activity is engaged rapidly through direct limbic and hypothalamic delivery, making the nasal spray format particularly well-suited to research protocols examining acute stress response modulation.
4. BDNF Upregulation and Neuroplasticity Support
Selank significantly upregulates brain-derived neurotrophic factor (BDNF) expression in hippocampal and cortical regions — a property that contributes to both its cognitive-enhancing and neuroprotective effects. BDNF is essential for long-term potentiation (LTP), the synaptic strengthening process that underlies memory formation and learning. By increasing BDNF in the very brain structures most directly reached by intranasal delivery — the olfactory bulb, hippocampus, and limbic cortex — Selank Nasal Spray delivers its neuroplasticity-supporting effects with maximum efficiency precisely where they are most relevant.
5. Immunomodulation and Immune-Brain Axis Research
Derived from the immunoactive tetrapeptide tuftsin, Selank retains its predecessor’s capacity to modulate immune function through cytokine regulation, macrophage activation, and T-lymphocyte modulation. Research has demonstrated Selank’s ability to normalise interleukin profiles dysregulated by chronic stress — a finding highly relevant to the growing field of psychoneuroimmunology, which studies the bidirectional communication between the brain and immune system. The intranasal route, with its direct access to brain-immune interface structures in the olfactory system, may offer unique advantages for studying this immune-brain axis modulation compared to peripheral injection.
6. Needle-Free Convenience for Extended Research Protocols
Beyond its pharmacokinetic advantages, the nasal spray format offers significant practical benefits for extended research protocols. The elimination of injections removes the requirement for sterile injection technique, reduces participant burden and compliance issues in human research studies, and avoids the local tissue reactions and injection site considerations associated with subcutaneous peptide administration. For research programmes involving repeated daily dosing over weeks or months, the nasal spray format substantially improves protocol adherence and participant experience — factors that are critical for the quality and reproducibility of longitudinal research data.
7. Mood Stabilisation and Emotional Regulation
Through its combined serotonergic, GABAergic, dopaminergic, and enkephalinase-inhibitory mechanisms, Selank produces a broad-spectrum mood-stabilising effect characterised by reduced emotional reactivity, improved frustration tolerance, decreased negative rumination, and enhanced overall sense of wellbeing. These mood effects are reported consistently in both clinical patient populations and research subject cohorts. The rapid central delivery of the nasal spray format means these mood-stabilising effects are available within minutes of administration, providing a uniquely fast-acting and non-pharmacologically burdensome approach to mood regulation research.
Selank Nasal Spray vs Selank Injectable — Which Is Right for Your Research?
Both formulations contain identical Selank heptapeptide but differ in delivery route, onset kinetics, and practical administration considerations:
- Onset Speed: Nasal spray produces central effects in 10–20 minutes via direct nose-to-brain transport; subcutaneous injection achieves systemic absorption in 15–30 minutes with subsequent BBB crossing required for central effects
- Central Bioavailability: Intranasal delivery achieves direct CNS delivery via olfactory and trigeminal pathways bypassing the BBB; subcutaneous injection relies on systemic circulation and BBB transport, which limits central peptide availability
- Administration Convenience: Nasal spray is needle-free, portable, and self-administrable without injection technique; injectable requires sterile reconstitution, syringe preparation, and injection competency
- Approved Clinical Format: The nasal spray is the approved pharmaceutical delivery route for Selank in Russia and Ukraine — the format validated in all clinical trials and with the longest clinical safety record
- Research Protocol Suitability: Nasal spray is preferred for longitudinal studies, human subject research, and protocols requiring rapid onset; injectable may be preferred for controlled dose-response studies requiring precise systemic pharmacokinetic characterisation
- Volume and Dose Precision: Metered nasal spray pumps deliver consistent per-actuation volumes; injectable allows finer dose titration through reconstitution concentration adjustments
Research Dosage and Administration Protocol
Selank Nasal Spray 10mg is administered intranasally using the metered pump spray device. Dosage is based on the approved Russian clinical protocol and published research data:
- Approved Clinical Dose: 400mcg per nostril (2 drops or 1 actuation of 0.15% solution), administered 2–3 times daily
- Standard Research Protocol: 1–2 actuations per nostril, 2–3 times daily, consistent with clinical trial dosing
- Cycle Length: 10–14 days per treatment course; repeat courses after 1–2 week rest period
- Administration Technique: Gently insert spray tip into nostril, tilt head slightly forward, actuate pump while inhaling gently — alternate nostrils per dose
- Onset of Effects: Anxiolytic effects typically apparent within 10–20 minutes; cognitive enhancement effects develop over the first few days of consistent use
- Storage: Store at 2–8°C; protect from light and heat; do not freeze; use within 30 days of opening
- Compatibility: Commonly researched alongside Semax nasal spray for complementary anxiolytic and cognitive enhancement protocols
Product Specifications
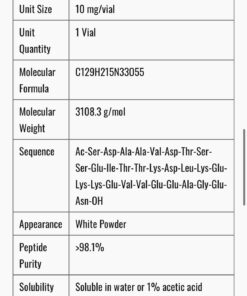
- Peptide Name: Selank (TP-7)
- Amino Acid Sequence: Thr-Lys-Pro-Arg-Pro-Gly-Pro
- CAS Number: 129954-34-3
- Total Peptide Content: 10mg per bottle
- Formulation: Sterile isotonic aqueous nasal spray solution
- Concentration: 0.15% w/v (1.5mg/ml) — consistent with approved clinical formulation
- Volume: Approximately 6.7ml per bottle
- Delivery Device: Metered-dose nasal spray pump
- Purity: ≥98% (HPLC and mass spectrometry verified)
- Preservative: Trace preservative system consistent with ophthalmic/nasal pharmaceutical standards
- Storage: 2–8°C; protect from light; do not freeze
- Intended Use: Research and laboratory purposes only
Why Choose Our Selank Nasal Spray 10mg?
Formulating Selank as a nasal spray introduces additional quality requirements beyond those of lyophilized injectables — the peptide must be dissolved in a sterile aqueous matrix at the correct concentration, the solution must be isotonic and pH-balanced for nasal mucosal compatibility, and the delivery device must provide consistent, reproducible metered dose actuation. Our Selank Nasal Spray 10mg is produced to meet all of these requirements. The peptide active ingredient is verified at ≥98% purity by independent HPLC and mass spectrometry before formulation, and the final product undergoes sterility testing and dose-per-actuation verification before batch release.
Our Quality and Formulation Standards
- ≥98% Selank peptide purity verified by independent third-party HPLC analysis
- Correct heptapeptide sequence (Thr-Lys-Pro-Arg-Pro-Gly-Pro) confirmed by mass spectrometry
- Sterile aqueous formulation at 0.15% concentration matching the approved clinical standard
- Isotonic, pH-balanced nasal spray solution for mucosal biocompatibility
- Consistent metered-dose pump delivering reliable per-actuation dose volume
- Sterility tested final product with tamper-evident sealed packaging and batch traceability
- Cold-chain shipping with temperature-protective packaging to maintain solution stability
- Discreet, professional worldwide delivery with knowledgeable customer support
Frequently Asked Questions About Selank Nasal Spray 10mg
Is the nasal spray more effective than the injectable form of Selank?
The nasal spray is the clinically validated and approved delivery format for Selank — it is the route used in all Russian clinical trials that established Selank’s efficacy and safety profile. From a central nervous system bioavailability perspective, intranasal delivery achieves direct nose-to-brain transport via olfactory and trigeminal pathways that bypass the blood-brain barrier entirely, potentially delivering higher brain concentrations than equivalent systemic doses. For most research applications — particularly those focused on anxiolytic effects, cognitive enhancement, and mood modulation — the nasal spray is considered the optimal and most authentic delivery format. The injectable form may be preferred for precise pharmacokinetic dose-response studies requiring controlled systemic exposure.
How many doses does a 10mg bottle provide?
At the standard approved clinical concentration of 0.15% (1.5mg/ml), a 10mg bottle contains approximately 6.7ml of solution. At a standard research dose of 400mcg (0.4mg) per administration — equivalent to one metered actuation per nostril per dose — the 10mg bottle provides approximately 16–20 full bilateral doses (both nostrils per administration). At 2 administrations daily, this provides approximately 8–10 days of research supply per bottle, consistent with the standard 10–14 day clinical treatment course for which Selank nasal spray is prescribed.
Can Selank Nasal Spray be stored at room temperature?
Selank Nasal Spray should be stored refrigerated at 2–8°C to maintain peptide stability and solution sterility throughout its shelf life. Unlike lyophilized peptide powders that can withstand room temperature storage for limited periods, an aqueous peptide solution is susceptible to hydrolytic degradation and microbial contamination at elevated temperatures. Brief periods at room temperature during normal use are acceptable, but the bottle should be returned to refrigerated storage promptly after each use session. The product should not be frozen, as freeze-thaw cycling can cause peptide aggregation and loss of activity in solution formulations.
2 reviews for Selank spray 10 mg
Add a review Cancel reply
Related products
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide











Ben –
This product has been a great addition to my regimen. The effects took a bit to kick in, but I’m happy with the results. Customer support was also responsive.
Brown –
Fantastic quality and fast shipping! I’ve been using this for a month and already see a difference. Highly recommend this to others.