Retatrutide For Sale
$185.00
Retatrutide 10mg — the next-generation triple agonist peptide targeting GLP-1, GIP, and glucagon receptors for superior fat loss and metabolic health. Lab-tested.
Retatrutide 10mg The Next-Generation Triple Agonist Peptide for Advanced Fat Loss, Metabolic Health, and Obesity Research
Retatrutide 10mg represents one of the most significant advances in metabolic peptide research to emerge in the last decade. As a triple receptor agonist — simultaneously activating glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic polypeptide (GIP), and glucagon receptors — Retatrutide operates across three complementary metabolic pathways at once, producing a magnitude of fat loss and metabolic improvement that surpasses currently available dual and single receptor agonists. Developed by Eli Lilly and currently in Phase 2 and Phase 3 clinical trial programmes, Retatrutide has generated exceptional data in early trials, with participants demonstrating weight reductions of up to 24% of total body weight — results that position it as potentially the most powerful anti-obesity peptide compound ever studied in clinical settings.
What Is Retatrutide 10mg?
Retatrutide (LY3437943) is a novel, long-acting acylated peptide engineered to function as a single-molecule triple agonist at three critical metabolic receptors: the GLP-1 receptor (GLP-1R), the GIP receptor (GIPR), and the glucagon receptor (GCGR). Each of these receptor systems plays a distinct and complementary role in the regulation of energy intake, glucose homeostasis, fat oxidation, and metabolic rate. By activating all three simultaneously through a single optimised molecule, Retatrutide produces a synergistic metabolic effect that no single or dual agonist can replicate.
The development of triple agonist compounds like Retatrutide represents the leading edge of incretin-based therapy research. Earlier generations of metabolic peptides — including semaglutide (GLP-1 mono-agonist) and tirzepatide (GLP-1/GIP dual agonist) — demonstrated progressively greater efficacy as additional receptor targets were incorporated. Retatrutide extends this progression by adding glucagon receptor agonism, which directly increases energy expenditure through thermogenesis and hepatic fat mobilisation — a mechanism absent from GLP-1 and GIP agonists alone.
Each vial of Retatrutide 10mg contains a precisely manufactured lyophilized dose of the peptide, produced under strict quality control standards to ensure purity, molecular integrity, and research-grade consistency.
Mechanism of Action — How Retatrutide Targets Three Metabolic Pathways
GLP-1 Receptor Agonism — Appetite Suppression and Glucose Control
Glucagon-like peptide-1 is an incretin hormone secreted by intestinal L-cells in response to food intake. GLP-1 receptor activation produces several complementary metabolic effects: it stimulates glucose-dependent insulin secretion from pancreatic beta cells, suppresses glucagon release, slows gastric emptying to prolong satiety, and acts on hypothalamic appetite centres to dramatically reduce hunger signals and caloric intake. These combined effects explain why GLP-1 receptor agonists were first recognised as anti-diabetic agents and subsequently identified as powerful weight loss compounds. Retatrutide’s GLP-1 component delivers the full spectrum of these appetite-suppressing and glycaemic-regulating effects as one pillar of its triple mechanism.
GIP Receptor Agonism — Enhanced Insulin Sensitivity and Fat Storage Regulation
Glucose-dependent insulinotropic polypeptide is secreted by intestinal K-cells and was originally identified as an incretin hormone that potentiates insulin secretion. More recent research has revealed that GIPR activation also plays critical roles in adipose tissue metabolism, bone turnover, and energy balance. GIPR agonism appears to enhance GLP-1 receptor signalling synergistically — the combination of GLP-1 and GIP activation produces greater weight loss and metabolic improvement than either alone. GIPR signalling in adipocytes also modulates fat storage dynamics and lipolysis, contributing to the preferential loss of visceral and hepatic fat observed in GIP/GLP-1 dual and triple agonist studies.
Glucagon Receptor Agonism — Thermogenesis and Hepatic Fat Mobilisation
The glucagon receptor component is what most fundamentally distinguishes Retatrutide from all dual and single agonist predecessors. Glucagon receptor activation increases hepatic glucose output, stimulates lipolysis in adipose tissue, enhances thermogenesis in brown adipose tissue (BAT), and accelerates fatty acid oxidation in the liver. In isolation, glucagon receptor agonism would raise blood glucose — an undesirable effect in metabolic disease. However, when balanced against simultaneous GLP-1 and GIP receptor activation — which powerfully suppress glucagon-driven glucose elevation through incretin effects — the net result is enhanced fat oxidation and energy expenditure without clinically meaningful hyperglycaemia. This elegant pharmacological balance is the engineering achievement at the heart of Retatrutide’s design and its key differentiator.
Key Benefits of Retatrutide 10mg
1. Unparalleled Weight Loss Magnitude
Phase 2 clinical trial data for Retatrutide published in The New England Journal of Medicine reported mean weight reductions of approximately 17.5% at 24 weeks and up to 24.2% at 48 weeks in participants receiving the highest dose. These figures represent the greatest weight loss magnitude ever recorded in a clinical trial of a pharmaceutical anti-obesity agent — surpassing semaglutide (approximately 15% at 68 weeks) and tirzepatide (approximately 22% at 72 weeks) in comparable timeframes. For research into the upper limits of pharmacologically achievable weight reduction, Retatrutide defines a new benchmark.
2. Superior Reduction of Visceral and Hepatic Fat
Retatrutide’s triple mechanism produces disproportionately large reductions in visceral adipose tissue (VAT) and intrahepatic fat — the metabolically active fat depots most strongly associated with insulin resistance, cardiovascular disease, type 2 diabetes, and non-alcoholic fatty liver disease (NAFLD). The glucagon receptor component specifically mobilises hepatic lipids, while GIP and GLP-1 components reduce lipogenic signalling and enhance peripheral fat clearance. Clinical imaging data from Phase 2 trials confirmed substantially greater reductions in liver fat content compared to placebo, with implications for NAFLD and NASH research applications.
3. Comprehensive Glycaemic Improvement
Through coordinated GLP-1 and GIP receptor activation, Retatrutide delivers robust improvements in all major markers of glycaemic control: fasting glucose, postprandial glucose excursions, HbA1c, and insulin sensitivity. In Phase 2 trial data, Retatrutide-treated participants demonstrated HbA1c reductions of up to 2.02 percentage points — a clinically significant improvement that rivals dedicated anti-diabetic medications. This positions Retatrutide as a compound of exceptional interest in type 2 diabetes research and cardiometabolic disease prevention studies.
4. Increased Energy Expenditure and Thermogenesis
Unlike GLP-1 mono-agonists whose weight loss effects are driven primarily by caloric restriction through appetite suppression, Retatrutide also increases energy expenditure through glucagon receptor-mediated thermogenesis in brown adipose tissue and enhanced fatty acid oxidation rates. This means Retatrutide addresses both sides of the energy balance equation — reducing caloric intake through appetite suppression while simultaneously increasing the rate at which the body burns existing fat stores. This dual-direction energy balance modulation is a fundamental pharmacological advantage over appetite-suppression-only mechanisms.
5. Cardiovascular and Cardiometabolic Benefits
Weight loss of the magnitude produced by Retatrutide carries profound secondary cardiovascular benefits, including reductions in blood pressure, improvement in lipid profiles (decreased triglycerides, LDL cholesterol, and total cholesterol), reduced systemic inflammation, and improved endothelial function. GLP-1 receptor agonists as a class have independently demonstrated direct cardioprotective effects beyond weight loss — including reduced major adverse cardiovascular events (MACE) in high-risk populations — and Retatrutide is expected to carry forward and potentially amplify these cardiometabolic benefits given its superior efficacy profile.
6. Preservation of Lean Muscle Mass
One concern with any aggressive weight loss intervention is the proportion of lean muscle mass lost alongside fat tissue. Retatrutide’s Phase 2 data indicated that the compound produced predominantly fat mass reduction with relative preservation of lean body mass — a finding consistent with the metabolic selectivity expected from a compound that primarily enhances fat oxidation and thermogenesis rather than inducing generalised catabolism. This lean mass preservation is clinically important for maintaining metabolic rate, physical function, and quality of life during weight loss.
Clinical Trial Data and Research Background
Phase 2 Trial Results — NEJM Publication
The landmark Phase 2 clinical trial of Retatrutide (NCT04881760) enrolled 338 adults with obesity or overweight with at least one weight-related comorbidity. Participants were randomised to receive weekly subcutaneous injections of Retatrutide at various doses (1mg, 4mg, 8mg, or 12mg) or placebo over 24 weeks, with extended follow-up to 48 weeks for a subset. The results, published in The New England Journal of Medicine in 2023, demonstrated dose-dependent weight loss across all active treatment groups, with the 12mg dose cohort achieving the unprecedented mean weight reduction of 24.2% at 48 weeks. No dose-limiting safety signals were identified beyond the gastrointestinal side effects typical of incretin-based therapies.
Ongoing Phase 3 Programme
Following the exceptional Phase 2 results, Eli Lilly initiated the TRIUMPH Phase 3 clinical trial programme to evaluate Retatrutide across obesity, type 2 diabetes, and cardiovascular disease indications. These trials are designed to confirm efficacy and safety at scale, assess long-term cardiovascular outcomes, and support regulatory submission for pharmaceutical approval. The progression of Retatrutide to Phase 3 represents a significant milestone in obesity pharmacotherapy and underscores the compound’s potential to become a transformative treatment option in metabolic medicine.
Retatrutide vs Semaglutide vs Tirzepatide — The Incretin Agonist Evolution
The incretin agonist landscape has advanced rapidly through successive generations of receptor targeting. Understanding where Retatrutide sits in this evolution clarifies its research significance:
- Semaglutide (Ozempic/Wegovy) — GLP-1 mono-agonist: Approximately 15% weight loss at 68 weeks (STEP trials). Gold standard first-generation GLP-1 therapy. No GIP or glucagon activity
- Tirzepatide (Mounjaro/Zepbound) — GLP-1/GIP dual agonist: Approximately 20-22% weight loss at 72 weeks (SURMOUNT trials). Adds GIP agonism for enhanced insulin sensitivity and fat metabolism
- Retatrutide — GLP-1/GIP/Glucagon triple agonist: Up to 24.2% weight loss at 48 weeks (Phase 2). Adds glucagon receptor agonism for thermogenesis and hepatic fat mobilisation — the most comprehensive metabolic coverage achieved by a single molecule to date
- Key differentiator: Each additional receptor target has produced progressively greater weight loss magnitude — Retatrutide’s glucagon component adds the thermogenic and lipolytic dimension that neither semaglutide nor tirzepatide can provide
- Safety profile: Retatrutide’s Phase 2 gastrointestinal adverse event profile (nausea, vomiting, diarrhoea) is qualitatively similar to other incretin agonists and dose-dependent — consistent with the known class effect
Research Dosage and Administration Protocols
Retatrutide 10mg is provided as a lyophilized powder for subcutaneous injection following reconstitution. The following information is derived from the published Phase 2 clinical trial protocol and researcher community data:
- Clinical Trial Doses Used: 1mg, 4mg, 8mg, and 12mg once weekly via subcutaneous injection
- Starting Dose (Escalation Protocol): Begin at 2mg once weekly; escalate gradually over 4-8 weeks to target dose to minimise GI side effects
- Target Research Dose: 4–8mg once weekly based on Phase 2 efficacy and tolerability data
- Reconstitution: Reconstitute lyophilized powder with bacteriostatic water; use 1–2ml per vial
- Storage: Lyophilized powder stable at -20°C for up to 24 months; refrigerate reconstituted solution at 2–8°C and use within 28 days
- Injection Site: Subcutaneous injection into abdomen, thigh, or upper arm — rotate sites weekly
- Cycle: Continuous weekly administration as per clinical trial protocol; dose escalation critical to GI tolerability
Product Specifications
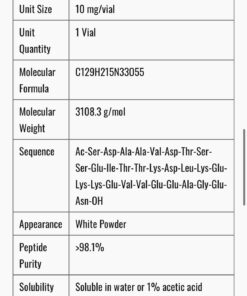
- Compound Name: Retatrutide (LY3437943)
- Type: Triple GLP-1/GIP/Glucagon receptor agonist peptide
- Vial Size: 10mg lyophilized powder
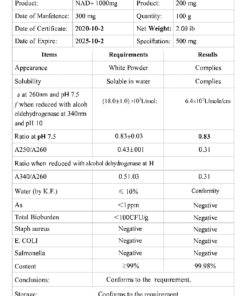
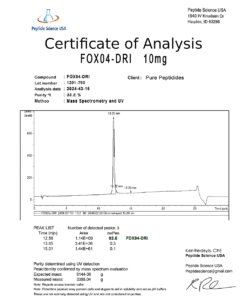
- Purity: ≥98% (HPLC and mass spectrometry verified)
- Appearance: White to off-white lyophilized powder
- Solubility: Soluble in bacteriostatic water or sterile saline
- Administration: Subcutaneous injection (post-reconstitution)
- Storage: -20°C lyophilized; 2–8°C reconstituted
- Shelf Life: 24 months lyophilized from manufacture date
- Intended Use: Research and laboratory purposes only
Why Choose Our Retatrutide 10mg?
As a next-generation research peptide with a complex molecular structure, Retatrutide demands the highest standards of synthesis quality and analytical verification. Our Retatrutide 10mg is produced using advanced solid-phase peptide synthesis with fatty acid acylation to replicate the long-acting pharmacokinetic profile of the clinical compound. Every batch undergoes independent third-party HPLC and mass spectrometry verification to confirm molecular identity, sequence accuracy, purity at 98% or above, and correct acylation — ensuring your research is built on a foundation of analytical certainty.
Our Quality Standards
- ≥98% purity independently verified by third-party HPLC chromatography
- Mass spectrometry confirmation of correct molecular identity and acylation profile
- Sterile filtration and lyophilization under GMP-aligned controlled manufacturing conditions
- Tamper-evident sealed vials with full batch traceability and Certificate of Analysis on request
- Cold-chain protective packaging maintaining peptide stability throughout international transit
- Discreet, professional worldwide shipping with temperature-protective materials
- Expert customer support team with in-depth knowledge of incretin peptide research
Frequently Asked Questions About Retatrutide 10mg
How does Retatrutide compare to tirzepatide in terms of weight loss?
Tirzepatide (a GLP-1/GIP dual agonist) achieved approximately 20 to 22% mean weight loss in the SURMOUNT-1 Phase 3 trial at 72 weeks — at the time the highest recorded weight loss in a pharmaceutical obesity trial. Retatrutide’s Phase 2 data demonstrated up to 24.2% mean weight loss at 48 weeks in the highest dose group — a shorter timeframe and a greater magnitude, driven by the addition of glucagon receptor agonism and its thermogenic and lipolytic effects. While cross-trial comparisons require caution, Retatrutide’s Phase 2 results represent a step-change improvement over tirzepatide’s benchmark figures.
What are the main side effects observed in Retatrutide clinical trials?
The adverse event profile of Retatrutide in Phase 2 trials was consistent with the well-characterised class effect of incretin-based therapies. The most commonly reported side effects were gastrointestinal in nature — nausea, vomiting, diarrhoea, and constipation — and were dose-dependent, most frequent during dose escalation, and generally transient. The incidence and severity of GI side effects were manageable with a gradual dose escalation protocol. No serious cardiovascular, hepatic, or renal safety signals were identified in Phase 2. Researchers should implement a structured dose titration schedule to optimise tolerability.
Is Retatrutide approved for human use?
As of the time of this writing, Retatrutide is not approved by the FDA, EMA, or any equivalent regulatory authority for therapeutic human use. It remains an investigational compound in active Phase 3 clinical development by Eli Lilly. This product is sold exclusively for research purposes. Researchers and institutions interested in Retatrutide’s clinical profile should monitor Eli Lilly’s ongoing TRIUMPH trial programme for the latest regulatory and efficacy data.
1 review for Retatrutide For Sale
Add a review Cancel reply
Related products
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide
Peptide














kelly benson –
Lost some reasonable lbs after using this highly recommended.