Uncategorized
The Role of Senescent Cells in Alzheimer’s Disease
The Role of Senescent cells in Alzheimer’s Disease
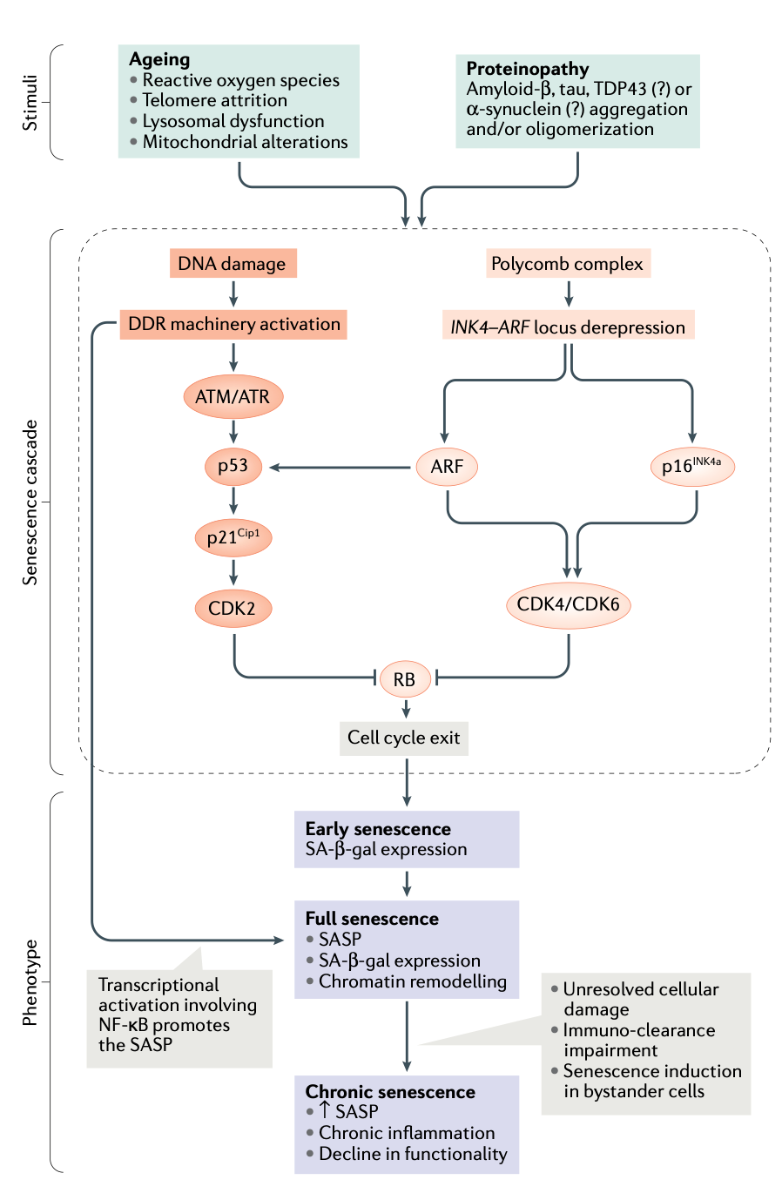
The aging process is strongly associated with developing diseases, such as cardiovascular conditions, hypertension, cancer, diabetes mellitus, osteoporosis, and neurodegenerative diseases, among others. [10,12] Alzheimer’s disease (AD) is no exception since aging is a risk factor for late-onset AD (more than 95% of AD cases). [14,15] Another factor highly associated with aging is the increased cellular senescence population of different cell types as we approach older ages (see Fig. 3).[14] Several studies suggest cellular senescence is critical in aging and connected conditions like AD.[2,5,10] Recent investigations have pointed out that senescent cells promote the pathogenesis of AD.[3-6] But what are senescent cells? Senescent cells’ particular feature is stopping the proliferation by entering a cell cycle arrest. [12-15] these senescent cells are also known to develop apoptosis resistance and secrete proinflammatory molecules.[11] Senescent cells not only remain even though they are “damaged” but also liberate various chemicals that can initiate inflammation.[3,7] Cellular senescence emerges when a cell receives considerable stress, driving it to “reprogram” its fate to an unlimited cell cycle arrest.[7,9] DNA damage, oncogene triggering, mitochondrial dysfunction, and the accumulation of proteins like the tau and the amyloid beta (Aβ) are well known to initiate senescence in different types of cells (see Fig. 1).[2,12,14]
Figure 1. The comparison between a healthy brain and an AD brain with senescent cells.[14]
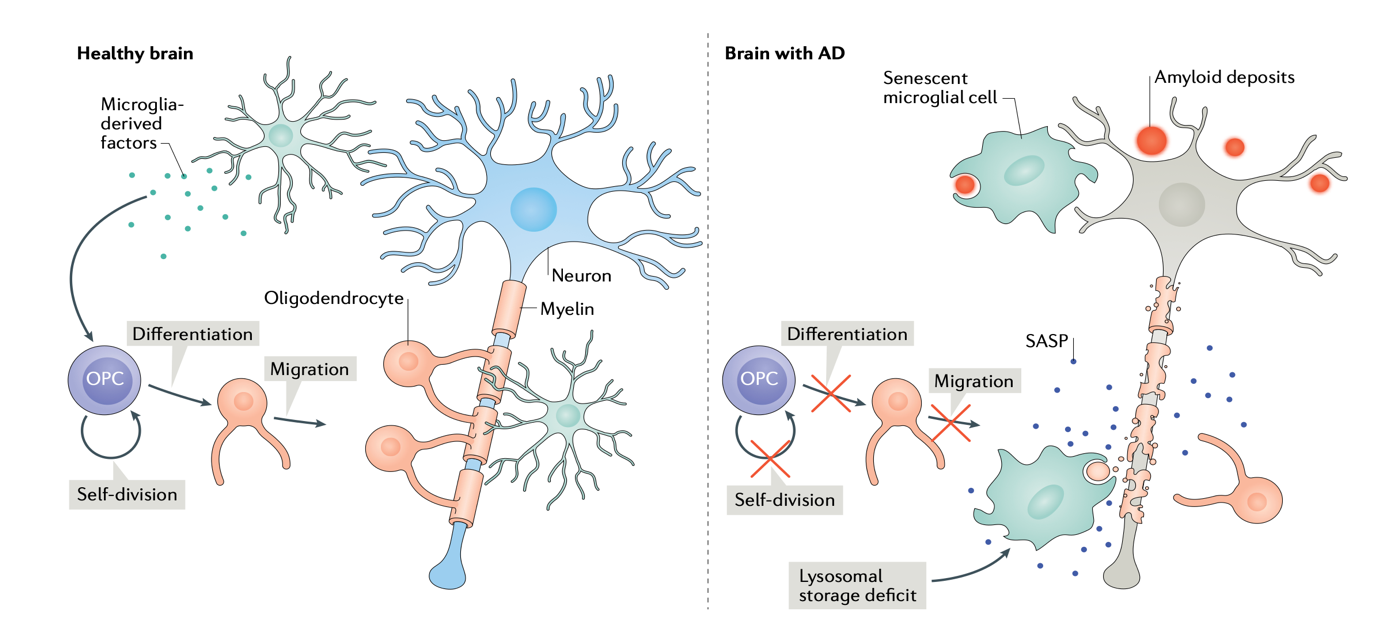
The formation of Aβ plaques in the extracellular area of neurons is a well-known hallmark of AD. [1,6] Various researchers recently identified that microglia and astrocytes tend to accumulate around the Aβ plaques already forming in AD brains. [8,13] Microglia and astrocytes are glial cells that maintain homeostasis and assist the neuron’s functioning. [3-5] Conversely, both cell types can activate the liberation of proinflammatory cytokines, which contribute to the chronic inflammation that characterizes AD. [3] Studies found that the astrocytes and microglia cells accumulated around the Aβ plaques show senescence characteristics.[8] Other studies demonstrated that the tau aggregation inside the cells triggers senescence in astrocytes and microglia.[1] In addition, the senescence state also has been identified in one of the most common cells in the brain, the oligodendrocyte progenitor cells (OPCs).[1,14] OPCs differentiate in mature oligodendrocytes, which are another type of glial cells.[5,6] The OPCs are activated when neuronal damage or demyelination is detected.[14] Demyelination is very closely related to AD since it is well known to contribute to axonal and neuron degeneration.[1,4] Several studies reveal that direct exposure to Aβ causes senescence in OPCs.[14] The constant exposition to Aβ and OPCs is also related to inducing senescence in other cell types.[3-6]
In recent years, researchers have focused most studies on the clearance of senescent cells as a medicinal strategy for AD. [12] A group of investigators cleared senescent glial and astrocyte cells from the brains of transgenic mice models, thus decreasing the formation of neurofibrillary tau tangles and increasing the cognition capacity of AD models.[2] In addition, other articles identified that the senescent cells expressed high levels of p16(Ink4a), which can be used as a biomarker for identifying senescence in cells.[4,14] Besides, they reported the increased expression of p16(Ink4a) in AD mice models before the formation of neurofibrillary tangles (see Fig. 3).[14] These findings highly suggest the relationship between senescent cells and the pathogenesis of AD. On the other hand, different investigations indicate that the accumulation of the tau protein, and not the Aβ plaques in transgenic mice models, induces a senescence phenotype in several cell types.[1] After using senolytics as a treatment method, they decreased the quantity of senescent cells in the brain of AD mouse models, thus reducing the formation of neurofibrillary tangles and increasing neuron survival. [1,11]
As a result of these findings, the use of senolytics to treat different conditions, including AD, has been increasing. [3-6] Senolytics are drugs that selectively aim to eliminate senescent cells.[4] There are several senolytics in clinical trials. Dasatinib and quercetin (D+Q). D+Q clears senescent OPCs and relieves Aβ plaque-associated inflammation and cognitive deficiencies in AD mice, suggesting a potential for senolytic treatment in AD. [6,13,15] D+Q is a promising therapy strategy for AD since several studies proved this drug to be safe, tolerable, and efficient in decreasing the senescent cells of the brain.[15] On the other hand, the FDA-approved Rapamycin is known to reduce the aggregation of Aβ and tau protein.[6,13,15] Rapamycin can improve the brain’s blood flow and enhance cognition in AD preclinical studies. The relationship between the pathogenesis and progression of AD with senescence is well-sustained and documented by several investigations, leading to the development of senolytics as a treatment for AD currently in clinical trials. [3-6, 15] Another essential aspect to consider is the genetic variants known to predispose people to AD. One of the genes is ADAMTS4, a component of senescent secretory phenotypes, and BIN1, which is triggered in response to genotoxic stress. [1,7,9] These genes strengthen the hypothesis of the relation between AD and senescence. In general terms, the participation of cellular senescence in the progression of AD is well documented. The relationship of cellular senescence with aging is critical to understanding the increased chances of developing a severe disorder as we approach older ages.
Figure 2. Results of treatment effects of D+Q in transgenic mice in comparison with a control group. MRI shows abnormal ventricles, cerebral blood flow (CBF), and white matter hyperintensity (WMH). The therapy alleviated the three symptoms.[13]

References
- Bryant, A. G., Hu, M., Carlyle, B. C., Arnold, S. E., Frosch, M. P., Das, S., … & Bennett, R. E. (2020). Cerebrovascular senescence is associated with tau pathology in Alzheimer’s disease.Frontiers in neurology, 11, 575953.
- Bussian, T. J. (2020). Cellular Senescence in Neurodegenerative Disease(Doctoral dissertation, College of Medicine-Mayo Clinic).
- Garbarino, V. R., Xu, P., Zhang, B., Biggs, G., Gonzales, M. M., & Orr, M. E. (2023). 45 Senolytic Therapy Transiently Reduces Inflammatory Markers in Primary Blood Mononuclear Cells of Individuals with Early Alzheimer’s Disease: Exploring the Conserved Transcriptional Response to Adversity as a Biomarker for Disease State.Journal of Clinical and Translational Science, 7(s1), 12-12.
- Gonzales, M. M., Garbarino, V. R., Kautz, T. F., Palavicini, J. P., Lopez-Cruzan, M., Dehkordi, S. K., … & Orr, M. E. (2023). Senolytic therapy in mild Alzheimer’s disease: a phase 1 feasibility trial.Nature Medicine, 29(10), 2481-2488.
- Gonzales, M. M., Garbarino, V. R., Zilli, E. M., Petersen, R. C., Kirkland, J. L., Tchkonia, T., … & Orr, M. E. (2022). Senolytic therapy to modulate the progression of Alzheimer’s disease (SToMP-AD): a pilot clinical trial.The journal of prevention of Alzheimer’s disease, 1-8.
- Gonzales, M. M., Krishnamurthy, S., Garbarino, V., Daeihagh, A. S., Gillispie, G. J., Deep, G., … & Orr, M. E. (2021). A geroscience motivated approach to treat Alzheimer’s disease: Senolytics move to clinical trials—mechanisms of ageing and development,200, 111589.
- Guerrero, A., De Strooper, B., & Arancibia-Cárcamo, I. L. (2021). Cellular senescence at the crossroads of inflammation and Alzheimer’s disease.Trends in Neurosciences, 44(9), 714-727.
- Han, X., Zhang, T., Liu, H., Mi, Y., & Gou, X. (2020). Astrocyte senescence and Alzheimer’s disease: A review.Frontiers in aging neuroscience, 12, 148.
- Herdy, J. R., Traxler, L., Agarwal, R. K., Karbacher, L., Schlachetzki, J. C., Boehnke, L., … & Gage, F. H. (2022). Increased post-mitotic senescence in aged human neurons is a pathological feature of Alzheimer’s disease.Cell Stem Cell, 29(12), 1637-1652.
- Liu, R. M. (2022). Aging, cellular senescence, and Alzheimer’s disease.International Journal of Molecular Sciences, 23(4), 1989.
- Maiese, K. (2023). The implications of telomere length: advanced aging, cell senescence, mri phenotypes, stem cells and alzheimer’s disease.Current Neurovascular Research, 20(2), 171-174.
- Martel, J., Ojcius, D. M., Wu, C. Y., Peng, H. H., Voisin, L., Perfettini, J. L., … & Young, J. D. (2020). Emerging use of senolytics and senomorphics against aging and chronic diseases.Medicinal research reviews, 40(6), 2114-2131.
- Riessland, M., Orr, M.E. Translating the Biology of Aging into New Therapeutics for Alzheimer’s Disease: Senolytics. J Prev Alzheimers Dis10, 633–646 (2023). https://doi.org/10.14283/jpad.2023.104
- Saez-Atienzar, S., & Masliah, E. (2020). Cellular senescence and Alzheimer disease: the egg and the chicken scenario.Nature Reviews Neuroscience, 21(8), 433-444.
- Zhang, P., Kishimoto, Y., Grammatikakis, I., Gottimukkala, K., Cutler, R. G., Zhang, S., … & Mattson, M. P. (2019). Senolytic therapy alleviates Aβ-associated oligodendrocyte progenitor cell senescence and cognitive deficits in an Alzheimer’s disease model.Nature neuroscience, 22(5), 719-728.
Appendix
Figure 3. Phenotypic features of senescent cells the cellular mechanism in neurodegeneration and aging