Uncategorized
PAN-AMPK Activator ATX-304 Improves Glucose Homeostasis and Microvascular Perfusion
Introduction to Type 2 Diabetes and the Emergence of ATX-304
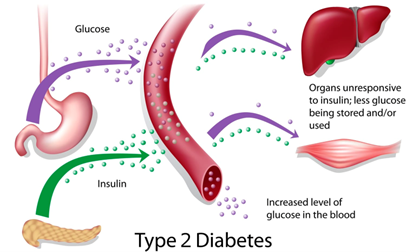 The United States is currently grappling with a severe Type 2 Diabetes (T2D) epidemic, a situation deeply intertwined with various lifestyle and demographic factors. This chronic condition, characterized primarily by insulin resistance and a progressive decline in insulin production, has become a major public health concern.
The United States is currently grappling with a severe Type 2 Diabetes (T2D) epidemic, a situation deeply intertwined with various lifestyle and demographic factors. This chronic condition, characterized primarily by insulin resistance and a progressive decline in insulin production, has become a major public health concern.
The latest data published in April 2023, the Centers for Disease Control and Prevention (CDC) reported that over 34 million Americans, which is approximately 10% of the U.S. population, suffer from diabetes. Most of these cases are Type 2 Diabetes.
Annually, the United States sees about 1.5 million new diagnoses of diabetes. This staggering number not only reflects the growing prevalence of the disease but also underscores the escalating public health challenge it poses. One of the most significant drivers of this epidemic is obesity, a condition that has become increasingly prevalent due to sedentary lifestyles and high-calorie diets. The link between obesity and T2D is well-established, with nearly 90% of individuals with Type 2 Diabetes being overweight or obese. This relationship highlights the need for targeted interventions focusing on lifestyle modifications as a key strategy in combating the diabetes epidemic.
The demographics of T2D in the U.S. further reveal that the condition affects certain groups more profoundly. It is more prevalent in older adults, with a higher incidence in individuals aged 45 and above. Additionally, there are significant disparities in terms of racial and ethnic backgrounds, with higher rates of diabetes observed in African American, Hispanic/Latino, Native American, and certain Asian populations compared to non-Hispanic whites.
Amidst these challenges, the development of ATX-304, a novel PAN-AMPK activator, marks a significant stride in diabetes research. This peptide has shown promising results in addressing the multifaceted complications of T2D.
AMP-Activated Protein Kinase (AMPK): The Target of ATX-304
AMP-Activated Protein Kinase (AMPK) is a critical enzyme in human physiology, often described as a master regulator of cellular energy homeostasis. Its central role is to maintain a balance of energy at both cellular and whole-body levels. AMPK operates as a sensor and regulator of energy status, activated in response to conditions where cellular energy levels are low, such as during exercise, stress, or periods of low glucose availability. The structure of AMPK is complex; it is a heterotrimeric enzyme consisting of three subunits – α, β, and γ. Each of these subunits plays a unique role in the enzyme’s activity and regulation.
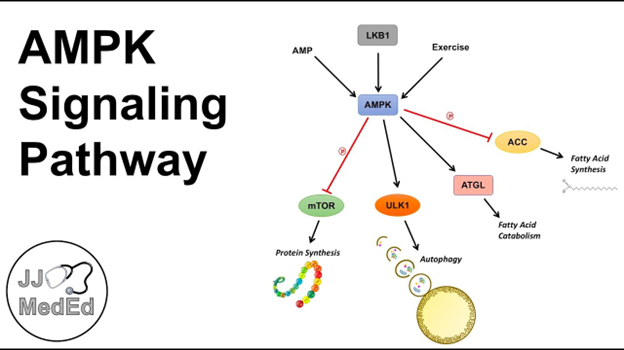
The activation of AMPK primarily occurs when there is a rise in the ratio of AMP (adenosine monophosphate) or ADP (adenosine diphosphate) to ATP (adenosine triphosphate) within cells, signaling a decrease in energy availability. As a cellular energy sensor, AMPK, upon activation, initiates a series of metabolic responses that lead to the generation of ATP, while simultaneously inhibiting energy-consuming processes that are not immediately vital for survival. The activation of AMPK has several key effects:
- Enhanced Glucose Uptake: AMPK plays a vital role in increasing glucose transport into cells, particularly muscle cells. This action is critical for boosting the availability of glucose, which is a primary substrate for energy production.
- Increased Fatty Acid Oxidation: The enzyme stimulates the breakdown of fats, providing an alternative energy source, especially important when glucose levels are low.
- Regulation of Protein Synthesis: AMPK inhibits protein synthesis, a process that typically consumes a significant amount of energy, thus conserving ATP for more critical functions.
- Modulation of Insulin Secretion: It also influences the functioning of pancreatic β cells, which are responsible for insulin production. This aspect is particularly relevant in the context of metabolic disorders like Type 2 Diabetes.
ATX-304, specifically designed as a PAN-AMPK activator, targets, and activates AMPK. This peptide drug mimics the natural physiological triggers of AMPK activation, such as exercise or caloric restriction. By activating AMPK, ATX-304 facilitates a series of metabolic actions within the body that are typically instigated by the enzyme under conditions of energy stress. This activation has several important implications:
- Improved Energy Regulation: ATX-304 aids in the efficient production and utilization of energy, particularly crucial in states of metabolic imbalance, such as in Type 2 Diabetes.
- Enhanced Glucose Homeostasis: The drug improves the body’s ability to regulate blood sugar levels, aiding in the reduction of hyperglycemia, a key issue in diabetes.
- Increased Metabolic Flexibility: By stimulating both glucose uptake and fatty acid oxidation, ATX-304 enhances the body’s metabolic flexibility, allowing for better adaptation to varying energy demands.
- Potential in Treating Metabolic Disorders: Given its role in improving energy balance and glucose metabolism, ATX-304 holds significant potential in the treatment of metabolic disorders, particularly Type 2 Diabetes.
In summary, to understand the mechanism of ATX-304, it’s crucial to comprehend the role of AMP-activated protein kinase (AMPK) in the body. AMPK serves as an energy sensor, playing a pivotal role in maintaining cellular energy homeostasis. It is activated under conditions of energy stress, such as exercise or caloric restriction, to facilitate the generation of ATP, the energy currency of the cell. Activation of AMPK leads to a cascade of metabolic adjustments, including increased glucose uptake, enhanced fatty acid oxidation, and modulation of insulin secretion.
ATX-304 is designed to target and activate AMPK, thereby harnessing its metabolic regulatory capabilities. By doing so, ATX-304 aims to emulate the beneficial effects of exercise and caloric restriction on glucose metabolism and energy balance.
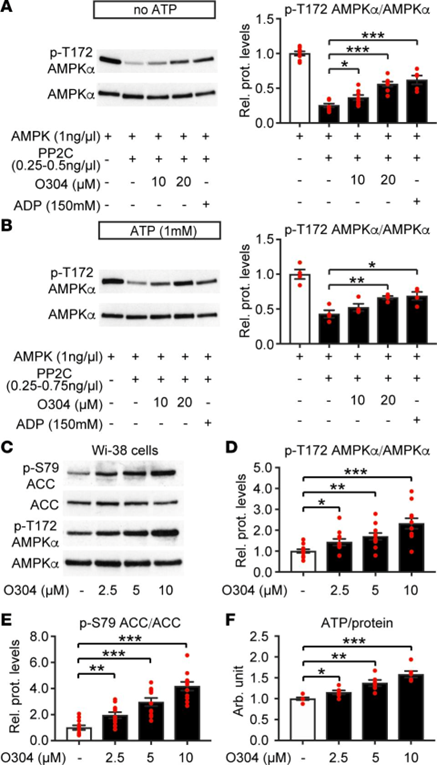
See the image below which shows ATX-304 increases p-T172 AMPK in vitro and increases p-T172 AMPK and ATP in cells.
(A and B) Representative immunoblot analysis and quantification of O304 dose-dependent suppression of PP2C-mediated dephosphorylation of p-T172 AMPK in absence (A) (n = 8 per condition) and presence (B) (n = 4 per condition) of 1.0 mM ATP. (C–E) Representative immunoblot analysis (C) and quantification of O304 dose-dependent increase of p-T172 AMPK (D) and p-S79 ACC (E) phosphorylation (n = 11 per condition) in Wi-38 human lung fibroblast cells. (F) Dose-dependent increase in ATP/protein levels in O304-treated Wi-38 human lung fibroblast cells (n = 6 per condition). Data are presented as mean ± SEM, *P < 0.05, **P < 0.01, ***P < 0.001 (Student’s t test).
The Impact of ATX-304 on Glucose Homeostasis
The impact of ATX-304 on glucose homeostasis is a critical aspect of its therapeutic profile, especially in the treatment of Type 2 Diabetes (T2D). Glucose homeostasis, the process of maintaining optimal blood glucose levels, is crucial for health. In T2D, this balance is disrupted due to insulin resistance, where cells don’t respond properly to insulin, and a decline in pancreatic β cell function, leading to reduced insulin production. This makes the regulation of blood glucose levels particularly challenging.
AMP-activated protein kinase (AMPK), the target of ATX-304, plays a significant role in glucose homeostasis. It promotes glucose uptake into cells, especially muscle cells, independently of insulin. This function is vital in managing T2D, where insulin resistance is a prevalent issue. Furthermore, AMPK activation can reduce glucose production in the liver, another key factor in controlling blood glucose levels.
ATX-304, as a PAN-AMPK activator, exerts its influence on glucose homeostasis through several mechanisms. Primarily, it stimulates AMPK, leading to enhanced glucose absorption into skeletal muscle cells. This action is critical in T2D, where overcoming insulin resistance is essential for effective treatment. By facilitating glucose uptake independently of insulin, ATX-304 can help lower blood glucose levels in a manner that bypasses the dysfunctional insulin signaling typically seen in T2D.
Moreover, ATX-304 may also impact hepatic glucose production through its activation of AMPK. By reducing the amount of glucose produced by the liver, it addresses another significant factor contributing to elevated blood glucose levels in diabetes. This dual action—enhancing glucose uptake in muscles and reducing hepatic glucose output—positions ATX-304 as a potentially powerful agent in restoring glucose balance in individuals with T2D.
In addition to these direct effects on glucose metabolism, ATX-304’s role in promoting β cell rest and reducing β cell stress indirectly contributes to improved glucose homeostasis. By alleviating the overburden on β cells, which are often overworked in T2D due to chronic high glucose levels, ATX-304 could help preserve or improve the insulin-producing capacity of these cells over time.
Enhancing Microvascular Perfusion with ATX-304
The enhancement of microvascular perfusion by ATX-304 is a significant aspect of its therapeutic potential, particularly in the context of Type 2 Diabetes (T2D). Microvascular complications are a common and severe consequence of T2D, characterized by damage to small blood vessels throughout the body. This can lead to various complications, such as neuropathy, retinopathy, and reduced tissue perfusion, which significantly impact the quality of life and disease prognosis in diabetic patients.
ATX-304’s role in improving microvascular perfusion is closely tied to its activation of AMP-activated protein kinase (AMPK). AMPK activation is known to promote vasodilation, the widening of blood vessels, which improves blood flow and, consequently, tissue perfusion. This action is particularly beneficial in T2D, where microvascular complications are a major concern. Improved blood flow in small vessels ensures better delivery of nutrients and oxygen to tissues, which is crucial for maintaining tissue health and function.
In the specific context of diabetes, enhanced microvascular perfusion can play a critical role in managing and potentially preventing some of the most debilitating complications of the disease. For instance, improved perfusion in peripheral tissues can help mitigate the risk of diabetic foot ulcers, a common issue in diabetic patients due to poor blood flow and nerve damage. Similarly, better blood flow to the retina can reduce the risk of diabetic retinopathy, a leading cause of blindness in diabetic patients.
Moreover, the improvement in microvascular blood flow can also enhance the muscle uptake of glucose and insulin, thereby aiding in better glycemic control. This is particularly relevant given that impaired microvascular perfusion in skeletal muscles is a contributing factor to insulin resistance, a hallmark of T2D.
ATX-304’s ability to enhance microvascular perfusion also has implications beyond direct diabetes management. By improving blood flow and tissue health, it can contribute to overall better health outcomes in patients with T2D, potentially reducing the risk of various complications and comorbidities associated with the disease.
ATX-304 and Cardiovascular Health
The relationship between ATX-304 and cardiovascular health is an integral component of its therapeutic value, particularly in the management of Type 2 Diabetes (T2D), a condition often accompanied by significant cardiovascular risks. Patients with T2D are at an increased risk of developing a range of cardiovascular diseases, including coronary artery disease, heart failure, and hypertension. The interplay between T2D and cardiovascular health is complex and multifactorial, often involving aspects like insulin resistance, chronic inflammation, and endothelial dysfunction.
ATX-304’s role in cardiovascular health primarily stems from its activation of AMP-activated protein kinase (AMPK), an enzyme that has widespread effects on cellular metabolism and energy balance. In the context of cardiovascular health, AMPK activation has been shown to have several beneficial effects. One of the key actions is the improvement of endothelial function, which is critical in maintaining vascular health. Endothelial cells line the blood vessels and play a crucial role in vascular tone and blood flow regulation. In T2D, endothelial function is often impaired, leading to reduced vasodilation, increased vascular stiffness, and a higher risk of atherosclerosis. By enhancing endothelial function, ATX-304 can help mitigate these cardiovascular risks.
Furthermore, ATX-304’s activation of AMPK also influences cardiac metabolism and function. In the diabetic heart, metabolic changes, such as impaired glucose uptake and increased fatty acid oxidation, contribute to cardiac dysfunction. ATX-304 can potentially reverse these metabolic abnormalities by increasing cardiac glucose uptake and improving overall cardiac efficiency. This action is particularly significant because it suggests that ATX-304 might help in preventing or reducing diabetic cardiomyopathy, a form of heart disease that develops in patients with T2D.
Another critical aspect of ATX-304’s impact on cardiovascular health is its potential to reduce blood pressure. Hypertension is a common comorbidity in patients with T2D and a major risk factor for cardiovascular events like heart attacks and strokes. The AMPK activation by ATX-304 could lead to relaxation of vascular smooth muscle, thus reducing vascular resistance and lowering blood pressure.
Moreover, the improvement in microvascular perfusion by ATX-304, as discussed earlier, also plays a role in cardiovascular health. Adequate perfusion ensures that cardiac and peripheral tissues receive sufficient blood supply, which is crucial for maintaining tissue health and function.
Clinical Implications and Future Directions
The journey of ATX-304 in research is a testament to the evolving landscape of diabetes treatment. Its multifaceted approach, targeting both glucose homeostasis and cardiovascular health, positions it as a potential game-changer in T2D therapy.
1. “Programs.” Www.amplifier-Tx.com, www.amplifier-tx.com/programs
