Uncategorized
miRNA Researched for Parkinson’s Disease: Neuroprotection Blocks Neuron Death
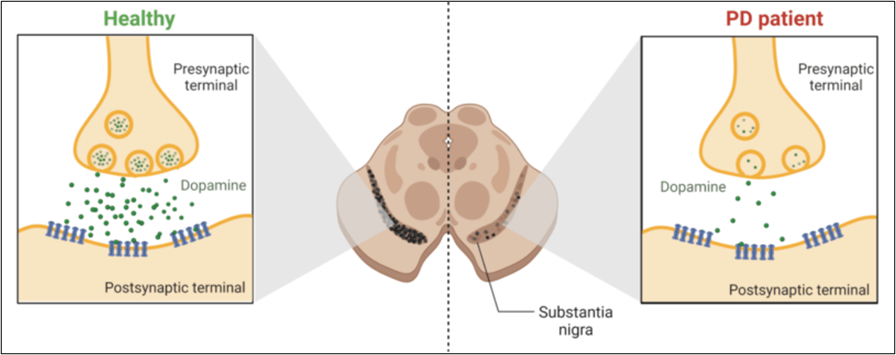
Programmed cell death, better known as apoptosis, is crucial in eliminating abnormal or unwanted cells in the body.[2] In recent years, the complex pathways regulating apoptosis have been well-studied as potential therapy targets.[2,10] Parkinson’s disease (PD) is no exception since the leading cause of this disorder is the death of dopamine-producing neurons in the brain (see Fig.1).[1-4] PD is a debilitating neurodegenerative disorder that steals a person’s ability to control movement.[8] Currently, levodopa (L-Dopa) is the most effective treatment for PD, helping in the improvement of the symptoms but not in the progression of the disease.[3] In addition, the use of L-Dopa led to adverse reactions after long-term administration.[3,7] Several studies propose using microRNAs (miRNAs) to inhibit the apoptosis of the dopamine-producing neurons found in PD.[6-9] miRNAs are tiny molecules capable of regulating gene expression (molecular switches) in the most critical processes for cell survival, like proliferation, cell differentiation, and apoptosis when required.[5-8]
Figure 1. The comparison between the normal functioning of dopaminergic neurons in healthy individuals vs in PD patients.
For this reason, understanding the role of miRNAs in PD could be vital to developing new treatments to decrease the progression of the disorder. The apoptosis process is significant for eliminating unwanted cells.[2] Therefore, using miRNAs as a therapy has the potential to inhibit unwanted cell death and induce apoptosis in abnormal cells as well.[3-6] Understanding this interplay between miRNAs and apoptosis could lead to new treatment strategies for PD.[7] Several investigations found that miRNAs have neuroprotection abilities, safeguarding neurons from apoptotic cell death.[5] Alternatively, miRNAs promote apoptosis, thus eliminating damaged or dysfunctional neurons, which is crucial to clear cellular debris from microenvironments.[5]
Some benefits of using miRNAs as a therapy for PD are the following: (1) miRNAs can target specific genes involved directly in the apoptotic pathway (specificity). (2) miRNAs are molecular switches, activating or inhibiting the apoptosis pathway. This adaptive characteristic is very convenient for this type of therapy since PD requires, in some cases, the inhibition of neuronal death (dopamine-producing neurons) and the activation of apoptosis in unwanted cells in the brain. (3) miRNAs can cross the blood-brain barrier and enter into cells, which is a challenge in developing neurodegenerative diseases. The apoptosis pathway accelerates the progression of PD by the direct dopamine depletion caused by the death of dopamine-producing neurons.[4-6] As the disease progresses, more and more neurons are lost, leading to worsening symptoms and disability.
miRNAs are tiny but powerful molecules that act like master regulators inside our cells.[1] They are non-coding RNA, meaning they do not directly code for proteins like most genes.[1,5] Instead, miRNAs adjust gene expression by targeting messenger RNA (mRNA), the intermediary molecule that carries instructions from DNA to build proteins.[5,9] Another interesting fact about miRNAs is their tumor suppressor capacity, as they target genes that promote cell growth and division.[1,3] Some studies have pointed out the presence of miRNAs in the cerebrospinal fluid and other body fluids like urine and blood, thanks to small vesicles called exosomes.[1,6] Some miRNAs are currently being investigated as possible biomarkers for PD, like miR-29c, miR-146a, miR-221, and miR-214.[5] The preliminary data suggest a significant difference between levels of these miRNAs in healthy vs. PD brains.[5]
Figure 2. miRNA used with different targets as a therapy for PD[5]
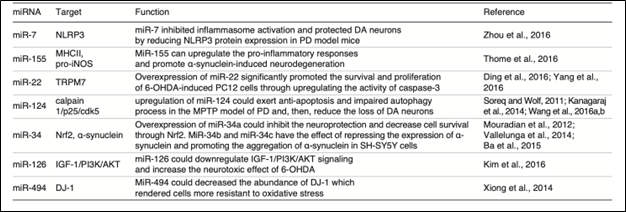
Another critical factor of neurodegeneration is the chronic neuroinflammation.[4] Several disorders, like PD and Alzheimer’s disease, among others, are well known to be affected by the overexpression and activation of microglia cells in the brain.[4,8] Microglia cells are the most abundant immune cells in the brain and are known to cause chronic inflammation in PD patients. Interestingly, several miRNAs are involved in the microglia inflammation response, like the MiR-155, which tends to upregulate proinflammatory responses and promote neurodegeneration caused by a-synuclein.[4] On the other hand, the overexpression of miR-22 supports neuronal survival and proliferation.[4,9] In general terms, miRNAs have been demonstrated to be a powerful tool for developing therapies against PD.[1-3] PD is a multifactorial disorder. The fact that miRNAs can target several genes simultaneously opens doors for potential therapeutic applications.[1,5] Apoptosis plays a crucial role in the development and progression of PD. Programmed cell death in dopaminergic neurons is considered the principal hallmark of PD, causing the loss of patients’ ability to control movement.[7,8] The results of the excessive loss of dopaminergic neurons are detrimental to the lifestyle of patients and their families.
Summary of the advantages of using miRNAs as a therapy against PD
- miRNAs are molecular switches capable of inhibiting or activating molecular processes as required.
- It can enter the blood-brain barrier and the cells quickly.
- Increases neuroplasticity
- Improves neuroprotection
- Promotes neuronal proliferation
- Served as biomarkers for PD
- They can be used as regulators for apoptosis, which plays a vital role in the development and progression of PD.
- It can be quickly delivered via vesicles with properties similar to those of exosomes.
- It helps decrease the over-activation of microglia cells inside the brain, thus reducing chronic neuroinflammation.
- It has the potential to decrease apoptosis in dopamine-producing neurons in the brain.
References
- Alieva, A. K., Filatova, E. V., Karabanov, A. V., Illarioshkin, S. N., Limborska, S. A., Shadrina, M. I., & Slominsky, P. A. (2015). miRNA expression is highly sensitive to a drug therapy in Parkinson’s disease. Parkinsonism & Related Disorders, 21(1), 72-74.
- Erekat, N. S. (2018). Apoptosis and its Role in Parkinson’s Disease. Exon Publications, 65-82.
- Leggio, L., Vivarelli, S., L’Episcopo, F., Tirolo, C., Caniglia, S., Testa, N., … & Iraci, N. (2017). microRNAs in Parkinson’s disease: from pathogenesis to novel diagnostic and therapeutic approaches. International journal of molecular sciences, 18(12), 2698.
- Li, S., Bi, G., Han, S., & Huang, R. (2022). MicroRNAs play a role in Parkinson’s disease by regulating microglia function: from pathogenetic involvement to therapeutic potential. Frontiers in Molecular Neuroscience, 14, 744942.
- Lu, X., Cui, Z., Liu, S., & Yin, F. (2018). MiRNAs participate in the diagnosis, pathogenesis and therapy of Parkinson’s disease.
- Ma, L., Wei, L., Wu, F., Hu, Z., Liu, Z., & Yuan, W. (2013). Advances with microRNAs in Parkinson’s disease research. Drug design, development and therapy, 1103-1113.
- Martinez, B., & Peplow, P. V. (2017). MicroRNAs in Parkinson’s disease and emerging therapeutic targets. Neural Regeneration Research, 12(12), 1945-1959.
- Mouradian, M. M. (2012). MicroRNAs in Parkinson’s disease. Neurobiology of disease, 46(2), 279-284.
- Titze-de-Almeida, S. S., Soto-Sánchez, C., Fernandez, E., Koprich, J. B., Brotchie, J. M., & Titze-de-Almeida, R. (2020). The promise and challenges of developing miRNA-based therapeutics for Parkinson’s disease. Cells, 9(4), 841.
- Yang, X., Zhang, M., Wei, M., Wang, A., Deng, Y., & Cao, H. (2020). MicroRNA-216a inhibits neuronal apoptosis in a cellular Parkinson’s disease model by targeting Bax. Metabolic Brain Disease, 35(4), 627-635.
