Uncategorized
How Does FOXO4-DRI Target Diabetes and Fibrosis?
Targeted Elimination of Senescent Beta Cells Prevents Type 1 Diabetes
“Pancreatic beta cells are usually thought as passive victims of immune cell attack in type 1 diabetes (T1D). Thompson et al. now demonstrate that a subpopulation of beta cells becomes senescent and actively promotes the immune-mediated destruction process. Clearance of senescent beta cells with small-molecule inhibitors prevents T1D.
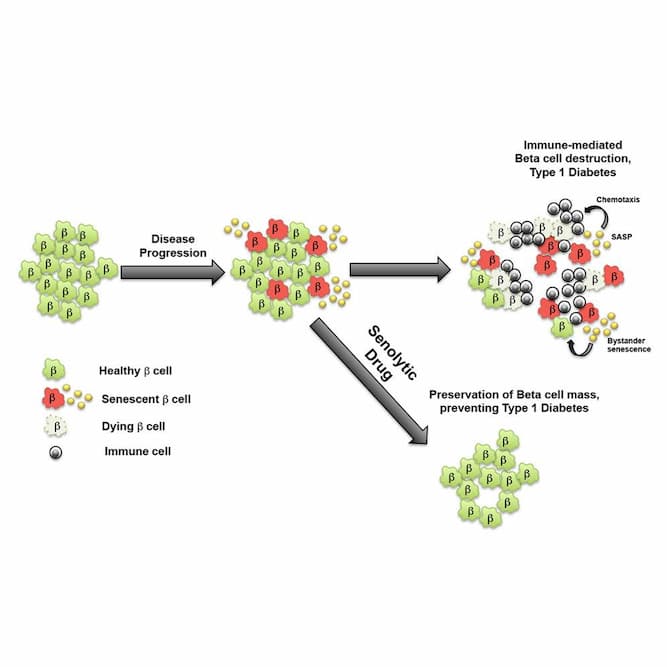
• Beta cells in NOD mice and human T1D become senescent
• Senescent beta cells acquire a senescence-associated secretory phenotype
• Senescent beta cells upregulate Bcl-2 and senolytic drugs induce their apoptosis
• Clearance of senescent beta cells preserves beta cell mass and prevents diabetes
Type 1 diabetes (T1D) is an organ-specific autoimmune disease characterized by hyperglycemia due to progressive loss of pancreatic beta cells. Immune-mediated beta cell destruction drives the disease, but whether beta cells actively participate in the pathogenesis remains unclear. Here, we show that during the natural history of T1D in humans and the non-obese diabetic (NOD) mouse model, a subset of beta cells acquires a senescence-associated secretory phenotype (SASP). Senescent beta cells upregulated pro-survival mediator Bcl-2, and treatment of NOD mice with Bcl-2 inhibitors selectively eliminated these cells without altering the abundance of the immune cell types involved in the disease. Significantly, elimination of senescent beta cells halted immune-mediated beta cell destruction and was sufficient to prevent diabetes. Our findings demonstrate that beta cell senescence is a significant component of the pathogenesis of T1D and indicate that clearance of senescent beta cells could be a new therapeutic approach for T1D.” (1)
“Clearance of Senescent Beta Cells with ABT-199 Prevents Diabetes“:
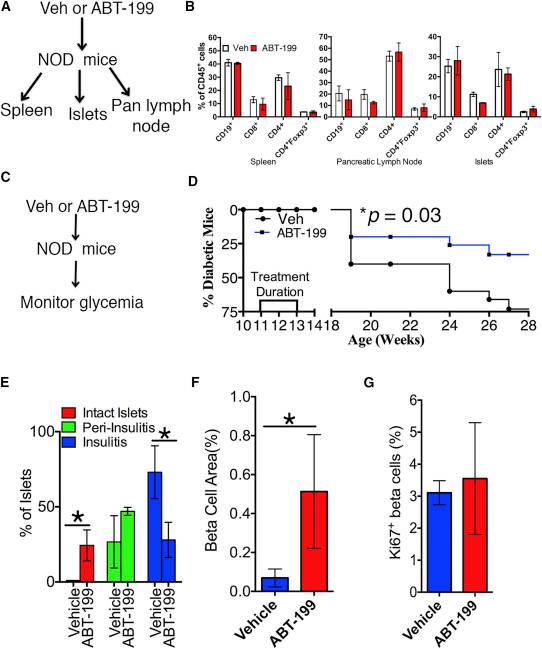
“Previous work has established that activation of the terminal unfolded protein response (UPR) in beta cells precedes symptoms of overt T1D. Indeed, inhibitors of the terminal UPR preserve beta cell mass and can reverse diabetes in the non-obese diabetic (NOD) mouse model, the classic model for spontaneous autoimmune diabetes, which recapitulates most of the features of T1D in humans. In addition to endoplasmic reticulum (ER) stress, beta cells are exposed to a variety of inflammatory cytokines from infiltrating lymphocytes. These factors are thought to trigger stress responses and apoptotic pathways, although the evidence for this mechanism in vivo is inconclusive. While it is generally accepted that apoptosis is the main response to terminal UPR, whether a beta cell mounts a protective or destructive stress response depends on the nature and duration of the stress as well as the competence of the beta cell to respond. Recent work has shown that intrinsic beta cell fragility is an underlying feature of both type 1 and 2 diabetes, prompting a closer investigation into the outcomes of the stress responses of beta cells in these diseases.
Here, we report that in the NOD mouse model and in human T1D, a subpopulation of beta cells undergoes a stress response leading to senescence and SASP. The secretory phenotype of these senescent beta cells exhibited non-cell-autonomous activities characteristic of SASP from other cell types, such as induction of paracrine senescence and stimulation of monocyte chemotaxis. We identified upregulation of Bcl-2 as the pro-survival mechanism of senescent beta cells and exploited this dependence using the small molecule BH3 mimetic known to preferentially trigger apoptosis in senescent cells, referred to as senolytic compounds. Elimination of senescent beta cells from NOD mice afforded robust protection against diabetes, indicating that this subpopulation of cells contributes to disease progression. Remarkably, senolytic treatment had no apparent effect on the major lymphoid or myeloid populations infiltrating the islets, in the spleen or pancreatic lymph nodes, suggesting that in these experiments ablation of senescent beta cells does not affect immune cells. Taken together, these findings demonstrate that SASP is a pathogenic mechanism in T1D and that targeted elimination of senescent beta cells prevents this disease.” (1)
“Beta Cells Undergo Stress-Induced Senescence during the Progression to T1D”:

Endothelial senescence: a new age in pulmonary hypertension
“Pulmonary hypertension is an enigmatic, deleterious disease driven by multiple heterogeneous causes with a burgeoning proportion of older patients with complex, chronic comorbidities without adequate treatment options. The underlying endothelial pathophenotypes that direct vasoconstriction and panvascular remodeling remain both controversial and incompletely defined. This review discusses emerging concepts centered on endothelial senescence in pulmonary vascular disease. This principle proposes a more heterogeneous, dynamic pulmonary endothelium in disease; it provides a potentially unifying feature of endothelial dysfunction in pulmonary hypertension irrespective of cause; and it supports a clinically relevant link between aging and pulmonary hypertension like other chronic illnesses.” (2)
FOXO4 peptide targets myofibroblast ameliorates bleomycin-induced pulmonary fibrosis in mice through ECM-receptor interaction pathway
“In this study, we explored the effect of FOXO4-DRI on bleomycin (BLM)-induced PF mouse model. We found that similar as the approved medication Pirfenidone, FOXO4-DRI decreased senescent cells, downregulated the expression of senescence-associated secretory phenotype (SASP) and attenuated BLM-induced morphological changes and collagen deposition. Furthermore, FOXO4-DRI could increase the percentage of type 2 alveolar epithelial cells (AEC2) and fibroblasts, and decrease the myofibroblasts in bleomycin (BLM)-induced PF mouse model.
Compared with mouse and human lung fibroblast cell lines, FOXO4‐DRI is inclined to kill TGF‐β‐induced myofibroblast in vitro. The inhibited effect of FOXO4‐DRI on myofibroblast lead to a downregulation of extracellular matrix (ECM) receptor interaction pathway in BLM‐induced PF. Above all, FOXO4‐DRI ameliorates BLM‐induced PF in mouse and may be served as a viable therapeutic option for PF.” (3)
“FOXO4‐D‐Retro‐Inverso(FOXO4‐DRI) ameliorates bleomycin (BLM)‐induced pulmonary fibrosis (PF).“:

“FOXO4‐DRI improves the impaired ratio of lung cells.”:

“FOXO4‐DRI works on the extracellular matrix (ECM) receptor interaction pathway to mitigate BLM‐induced PF… FOXO4‐DRI downregulates expression of proteins in ECM (extracellular matrix) receptor interaction.”:

“FOXO4‐DRI alleviates myofibroblast differentiation in BLM‐induced PF mouse.“:

“FOXO4‐DRI is inclined to kill TGF‐β‐induced myofibroblasts in vitro.“:


“In this study, we treated the BLM‐induced PF mouse model with FOXO4‐DRI, and found that besides the elimination of senescent cells and SASP, FOXO4‐DRI could attenuate BLM‐induced morphological changes and collagen deposition to the similar as the approved medication PFD. Furthermore, FOXO4‐DRI increases the percentage of type 2 alveolar epithelial cells (AEC2) and fibroblasts and decreases the myofibroblasts in BLM‐induced PF mouse models. Compared to mouse and human lung fibroblast cell lines, FOXO4‐DRI is inclined to kill TGF‐β‐induced myofibroblast in vitro. The inhibited effect of FOXO4‐DRI on myofibroblast and ECM proteins lead to a downregulation of ECM‐receptor interaction pathway in BLM‐induced PF. Above all, FOXO4‐DRI ameliorates BLM‐induced PF in mouse and may be served as a viable therapeutic option for PF.
Activation of fibroblast‐to‐myofibroblasts differentiation is one of the most important factor to deposit ECM. In this study, FOXO4‐DRI is more inclined to kill the myofibroblasts both in vivo and in vitro. The upregulation expression of FOXO4 protein in TGF‐β‐stimulated cells may provide the possible reason for the effect of FOXO4‐DRI. The formation of ECM also requires the secretion of ECM proteins. ECM is achieved by following a strict layered assembly pattern, which begins with the deposition of fibronectin filaments on the cell surface, a process called fibrillary formation. In this study, FOXO4‐DRI downregulated the main ECM protein significantly, which mainly contributed for the reducing of PF degree. Therefore, we summarize the whole process of FOXO4‐DRI works on BLM‐induced PF as follows, FOXO4‐DRI eliminated senescent cells, downregulation of SASP released by senescent cells reduce the inflammatory stimulation to neighbour cells to rescue BLM‐induced PF; On the other hand, FOXO4‐DRI is more inclined to kill myofibroblasts, downregulated the expression of main ECM proteins, reduced ECM formation and finally inhibited ECM‐receptor interaction to mitigate BLM‐induced PF.
PFD has been approved in Europe in 2011 for the treatment of IPF and in the USA in 2014. The recommended daily maintenance dose of PFD is 801 mg three times per day (2403 mg/day) with a 14‐day titration. Consistently with our results, it has been reported that PFD could ameliorate BLM‐Induced PF at administration of 300 mg/kg PFD for 3–4 weeks immediately after BLM treatment. Obviously, PFD needs to be given at very early time point in mouse, and the medication period is long. Compared with PFD, BLM‐induced lung fibrosis mouse was treated with 5 mg/kg FOXO4‐DRI for 3 times, started from 14 th day after BLM treatment; so the advantage over PFD, FOXO4‐DRI lies in its short administration time and the low treatment dose. Importantly, the same as other senolytic agents, it can be used to treat PF even when pathological changes have occurred. FOXO4‐DRI is a cell‐penetrating peptide, which can in theory target any surface‐exposed stretch of amino acids to block specific protein‐protein interactions, selectively modulate specific signalling events. Thus it have the advantages of target specificity and low toxicity over the reported senolytic agents. In addition, in the treatment of PF, FOXO4‐DRI may reduce the risk of cancer progression and metastasis by eliminating senescent cells, which have been demonstrated to contribute to cancer metastasis and relapse. At present, mouse was administrated with FOXO4‐DRI by intraperitoneal injection, it will be better to be improved as an oral gavage peptide. Above all, FOXO4‐DRI has great potential as a candidate for PF therapy.” (3)
Ageing, cellular senescence and chronic kidney disease: experimental evidence
“Kidney ageing and CKD are featured with increased cellular senescence, an irreversible state of cell cycle arrest and the cessation of cell division. Senescent cells secrete a diverse array of proinflammatory and profibrotic factors known as senescence-associated secretory phenotype (SASP). Secondary senescence can be induced by primary senescent cells via a mechanism involving direct contact or the SASP. Various senolytic therapies aiming to selectively remove senescent cells in vivo have been developed. Senostatic approaches to suppress senescence or inhibit SASP, as well as nutrient signalling regulators are also validated in animal models of ageing.
These recent studies provide experimental evidence supporting the notion that accumulation of senescent cells and their associated SASP is a main driver leading to structural and functional organ degeneration in CKD and other ageing-related disorder.” (4)
Resources
Product available for research use only:

