Uncategorized
UB-312 Parkinson’s Peptide Produces Antibodies Targeting α-synuclein Lewy bodies
UB-312 synthetic peptide induces antibodies against a-synuclein as a treatment for Parkinson’s disease.
Alpha-synuclein (αSyn) is an essential neuronal protein in the nerve terminal that inhibits neurotransmitter release when over-expressed.[8] In general terms, the αSyn preserves the normal function of synapses, which is how information is transmitted between neurons.[8] However, dysfunction of the αSyn protein causes the development of neuropathology. In recent years, the term synucleinopathies has been used to identify progressive neurodegenerative disorders caused by the aggregation of αSyn in the brain.[4] One of the most common synucleinopathy is Parkinson’s disease (PD). [4,8] Even though PD is the second most common neurodegenerative disorder worldwide, there is no cure. However, several investigations have found that αSyn accumulation is critical for the progression of the disease. [4-6] Recent studies show that targeting the assembly of αSyn in the brain slows down or even reverses the neurodegeneration in PD brain patients. [2-7] One of the most significant challenges investigators confront when developing new therapies for combating PD is the high selectiveness of the blood-brain barrier (BBB).[1] Even though it was believed that the BBB disrupts its integrity as soon as neurodegeneration starts, recent studies show that the BBB is so discriminating that almost all proteins and antibodies from the peripheral blood are restricted.[1] In other words, they can’t cross the BBB even with progressive neurodegeneration. This restriction has limited the development of traditional immunotherapy for PD and other related disorders.
Nevertheless, with the help of new technology, investigators have developed some very targeted vaccines capable of crossing the BBB to treat synucleinopathies. [6-8] UB-312 is a fully synthetic αSyn peptide conjugated with a T-helper peptide capable of inducing antibodies for combating the αSyn aggregation in the brain.[8] This synthetic peptide has been demonstrated to have great potential as an immunotherapy for PD (see Fig 1).[3] UB-312 is currently in clinical trials and is being tested on humans for the first time.[8] In a recent study where at least 23 participants received three UB-312 doses, they only presented minor side effects like fatigue, headache, vaccination-site discomfort, nasopharyngitis, and lumbar puncture-site pain.[8] Several studies suggest that UB-312 vaccine therapy is safe and well-accepted in vivo studies (including humans).[6-8] The most remarkable characteristic of the UB-312 treatment is the capacity to induce the production of antibodies for combating oligomeric and fibrillar forms of αSyn, thus decreasing or preventing the accumulation of αSyn in the brain (see Fig.2).[8] By causing the production of antibodies for combating the αSyn, the PD pathological progression can be slowed down or reverted.[2] Still, also this therapy can be used for treating other synucleinopathies like dementia with Lewy bodies and multiple system atrophy.[2-5]
The passive immunization pathway allows the immune system to recognize different sites of the αSyn, activating phagocytosis, thus decreasing levels of the αSyn protein in the brain.[3]
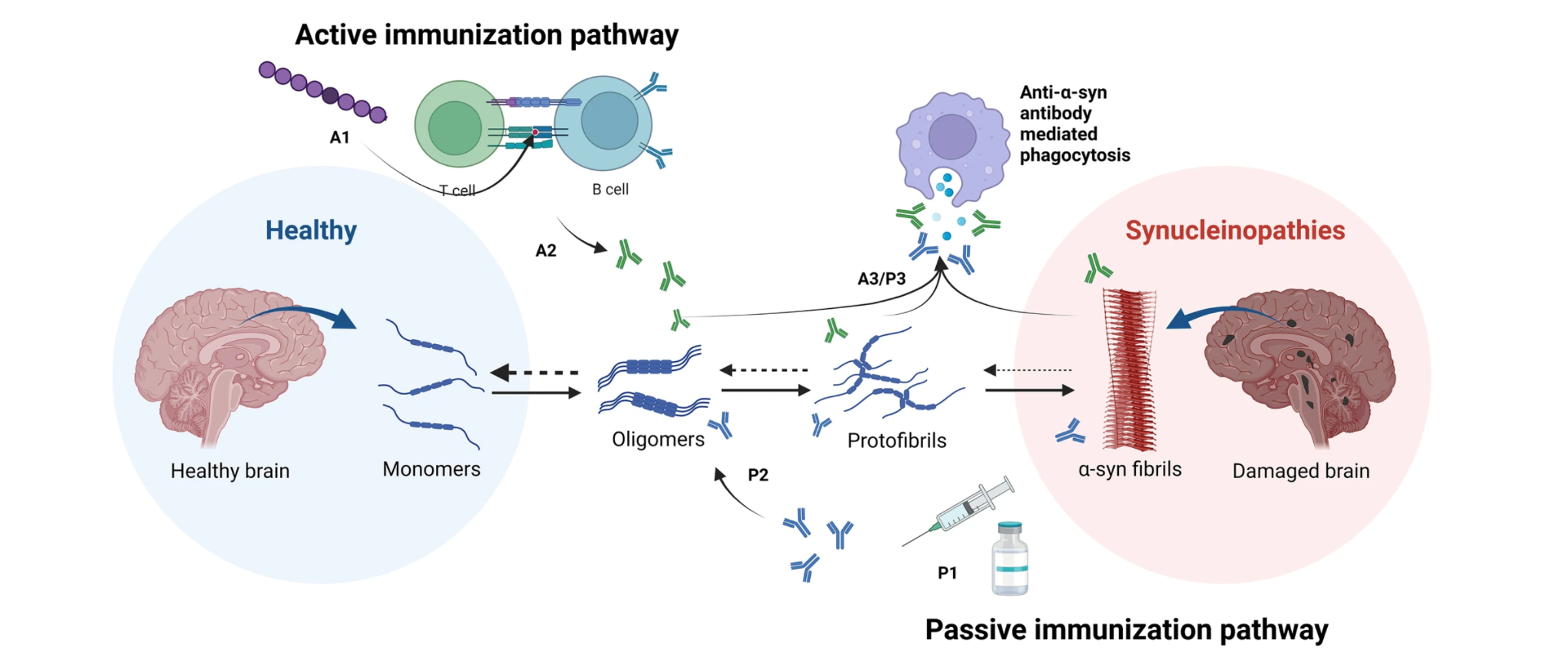
Figure 1.
The mechanism of the UB-312 is to target the 12 amino acid sequence in the C terminal of αSyn via the passive immunization pathway. [3,8] Some studies using UB-312 in guinea pigs induced the production of antibodies capable of binding the αSyn oligomers and fibrils of post-mortem brain from PD patients.[3] Oligomers and fibrils are different types of αSyn existing in the brain. The advantage of UB-312 is that the antibodies produced can attach to many forms of αSyn, avoiding the formation of fibrils and eliminating the already-formed fibrils (the most toxic structure of αSyn). [7,8] This strategy is a game-changer for PD pathology.
Cerebrospinal fluid and serum analysis for αSyn antibodies. The red arrows indicate the administration of a higher UB-312 dose.[8]
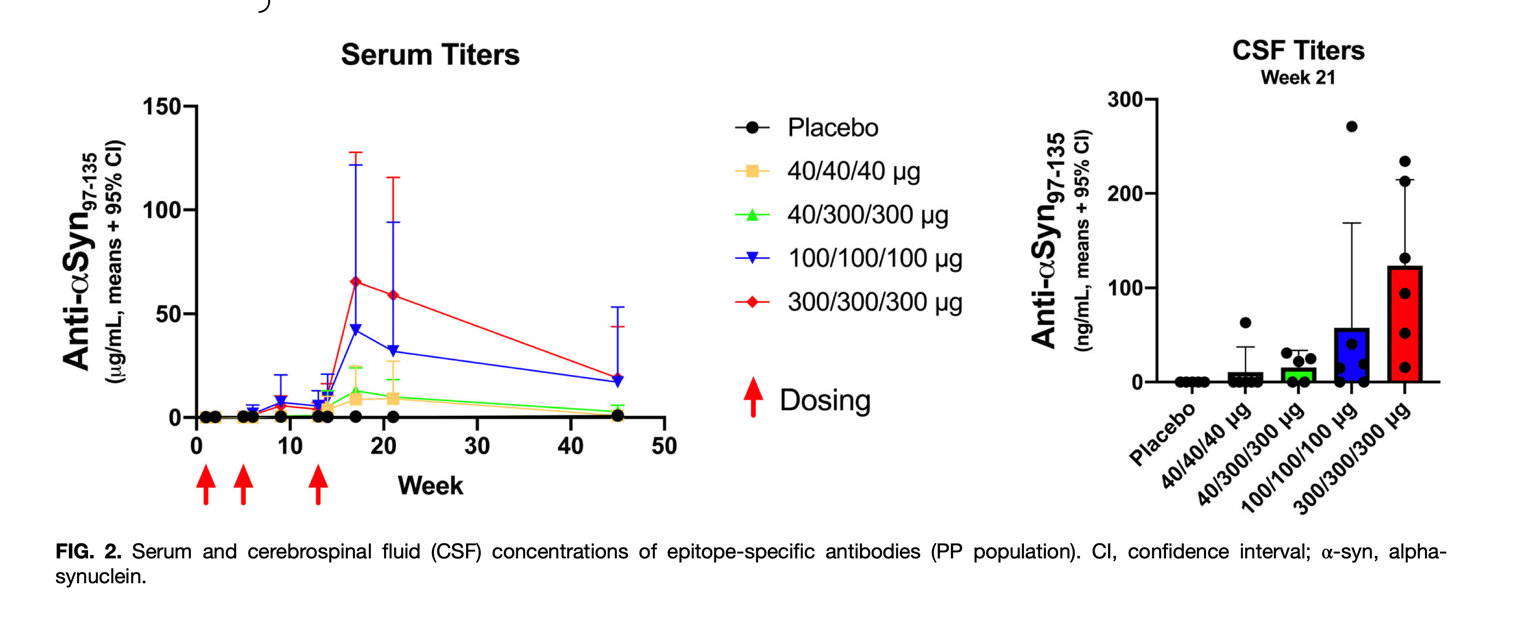
Figure 2.
Several investigations testing the UB-312 in mice show promising results in antibody production and improving motor performance.[7] Also, this synthetic peptide decreases αSyn oligomers in the brain according to several quantitative analyses.[7] Additionally, the UB-312 improves the local immune response in the brain.[7,8] It is essential to mention that UB-312 does not cause a T-cell infiltration to the brain.[2] This is done by inducing antibodies that are later transported to the brain, where they recognize a fragment of the αSyn, thus decreasing the accumulation of oligomers and the development of αSyn fibrils.[6-8] In general terms, one of the problems of immunotherapies is that the excessive inflammatory response can be detrimental to brain cells.[1,2] The immune system uses the inflammatory response in normal ranges for combating infections and disease.[5] However, in synucleinopathies like PD, the immune system activates the inflammatory response to “decrease” or combat the accumulation of αSyn.[5,8] However, the undeserved activation of the inflammatory response induces apoptosis (programming cell death). An incredible advantage of UB-312 is that it was created to avoid an exaggerated inflammatory response while triggering a humoral immune response.[7] Some other articles evaluated the αSyn reduction in the brain and the gastrointestinal system, finding a remarkable decrease in both.[5] UB-312 is the first clinical trial immunotherapy that has shown promising results in slowing down or reverting the progression of PD by targeting the αSyn oligomers and fibrils with antibodies.[5]
The capacity of this vaccine to target different forms of αSyn opens a wide range of opportunities for treating PD in various stages, including the early ones, which can help slow down or prevent the progression of PD. In addition, the few promising results in vivo models showing the improvement of motor performance demonstrated that UB-312 is worth studying. [2-8] In the preliminary data from the current clinical trials, satisfactory results have been reported in the induction of antibodies for combating the αSyn. [8] When comparing the levels of antibodies for αSyn before and after the vaccine, the results show a significant increase after three doses.[8] However, the investigators still evaluate the dose needed for the best performance. In general, the human trials demonstrated that UB-312 is well tolerated and dose-dependent for producing considerable quantities of the antibodies. [5,6,8] More studies need to be performed to evaluate the effectiveness of UB-312 in PD patients since the human trial was conducted in healthy individuals.
References
- Fleming, S. M., Davis, A., & Simons, E. (2022). Targeting alpha-synuclein via the immune system in Parkinson’s disease: Current vaccine therapies.Neuropharmacology, 202, 108870.
- Henriquez, G., & Narayan, M. (2023). Targeting α-synuclein aggregation with immunotherapy: a promising therapeutic approach for Parkinson’s disease.Exploration of Neuroprotective Therapy, 3(4), 207-234.
- Knecht, L., Folke, J., Dodel, R., Ross, J. A., & Albus, A. (2022). Alpha-synuclein immunization strategies for synucleinopathies in clinical studies: a biological perspective.Neurotherapeutics, 19(5), 1489-1502.
- Li, J., Luo, H., Zheng, H., Duan, S., Zhao, T., Yuan, Y., … & Xu, Y. (2022). Clinical application of prion-like seeding in α-synucleinopathies: Early and non-invasive diagnosis and therapeutic development.Frontiers in Molecular Neuroscience, 15, 975619.
- Nimmo, J. T., Smith, H., Wang, C. Y., Teeling, J. L., Nicoll, J. A., Verma, A., … & Carare, R. O. (2022). Immunization with UB-312 in the Thy1SNCA mouse prevents motor performance deficits and oligomeric α-synuclein accumulation in the brain and gut.Acta Neuropathologica, 143(1), 55-73.
- Nimmo, J. T., Verma, A., Dodart, J. C., Wang, C. Y., Savistchenko, J., Melki, R., … & Nicoll, J. A. (2020). Novel antibodies detect additional α-synuclein pathology in synucleinopathies: potential development for immunotherapy.Alzheimer’s Research & Therapy, 12, 1-16.
- Nimmo, J., Smith, H., Teeling, J., Nicoll, J., Verma, A., Dodart, J. C., … & Carare, R. O. (2021). Immunotherapy with UB-312 in the Thy1-SNCA mouse improves motor performance and reduces oligomeric α-synuclein in the brain and gut.Acta Neuropathologica.
- Yu, H. J., Thijssen, E., van Brummelen, E., van Der Plas, J. L., Radanovic, I., Moerland, M., … & Dodart, J. C. (2022). A Randomized First‐in‐Human Study With UB‐312, a UBITh® α‐Synuclein Peptide Vaccine.Movement Disorders, 37(7), 1416-1424
