Uncategorized
What is JNJ-2113 and How Does it Work?
About JNJ-77242113 (JNJ-2113)
JNJ-2113 stands out as the first oral peptide specifically designed to inhibit the IL-23 receptor. This means it blocks the activity of the IL-23 receptor, which is a protein involved in the immune system’s inflammatory processes. Interleukin-23 plays a significant role in the development and progression of plaque psoriasis, a chronic skin condition characterized by red, itchy, and scaly patches. By inhibiting the IL-23 receptor, JNJ-2113 can potentially reduce the inflammation, proliferation, and development of psoriatic plaques, thereby alleviating symptoms associated with moderate-to-severe plaque psoriasis.
In terms of its contribution to medicine, JNJ-2113 represents a novel approach to psoriasis treatment, especially for patients who might prefer or require an oral medication instead of the more commonly used injectable therapies. Its development is significant because it targets a specific pathway (IL-23 signaling) involved in the pathogenesis of psoriasis, which could lead to improved outcomes for patients with fewer side effects compared to broader immunosuppressive medications. If successful, JNJ-2113 could offer a new, potentially more convenient treatment option for those suffering from plaque psoriasis, improving their quality of life and easing the physical and psychological burden of the disease.
About Psoriasis
In America, psoriasis affects over 8 million people, representing a significant portion of the population. While there is no cure, many people with psoriasis can manage their symptoms effectively with medical treatment and lifestyle adjustments.
Psoriasis is a chronic autoimmune condition that primarily affects the skin, characterized by the rapid buildup of skin cells leading to thick, red, scaly patches that are often itchy or painful. This acceleration of skin cell growth is due to an overactive immune response. There are several forms of psoriasis, with plaque psoriasis being the most common, marked by raised, inflamed, red lesions covered with a silvery white buildup of dead skin cells. Other forms include guttate psoriasis, which appears as small, dot-like lesions; pustular psoriasis, characterized by white pustules surrounded by red skin; inverse psoriasis, which affects skin folds; and erythrodermic psoriasis, a severe form that leads to widespread redness over most of the body.
Treatments for psoriasis vary based on the type and severity of the condition and can include topical treatments, phototherapy, systemic medications, and biologic drugs, which target specific parts of the immune system. The goal of treatment is to reduce inflammation and clear the skin.
Mechanism of Action
The mechanism of action of JNJ-2113, a targeted oral peptide, involves specifically blocking the activity of the interleukin-23 (IL-23) receptor, which plays a critical role in the immune system’s inflammatory processes.
Here’s how it works:
Interleukin-23 (IL-23) Role: IL-23 is a cytokine, a type of signaling protein involved in regulating the immune system’s response. It is particularly important in the development and maintenance of chronic inflammation and autoimmunity, as seen in plaque psoriasis. IL-23 helps in the differentiation, survival, and activation of T cells, particularly a subset known as Th17 cells.
Th17 Cells and Inflammation: Once activated by IL-23, Th17 cells produce other inflammatory molecules, such as interleukin-17 (IL-17). These molecules contribute to the inflammation, skin cell proliferation, and other symptoms characteristic of plaque psoriasis.
Blocking IL-23 Receptor: JNJ-2113 is designed to bind directly to the IL-23 receptor on the surface of T cells and other relevant immune cells. By binding to this receptor, JNJ-2113 prevents IL-23 from interacting with its receptor, effectively blocking the downstream signaling pathway.
Reduction in Inflammation and Symptoms: By inhibiting the action of IL-23, JNJ-2113 reduces the activation of Th17 cells and the subsequent release of inflammatory molecules like IL-17. This leads to a decrease in inflammation, a reduction in the formation of psoriatic plaques, and relief from symptoms for individuals with moderate-to-severe plaque psoriasis.
In summary, JNJ-2113’s mechanism of action targets a specific part of the immune system’s inflammatory response, which is crucial in the pathogenesis of plaque psoriasis. By specifically blocking the IL-23 receptor, it aims to provide therapeutic effects with potentially fewer side effects compared to treatments that broadly suppress the immune system. This targeted approach represents a promising advancement in the treatment of plaque psoriasis and potentially other IL-23-mediated diseases.
JNJ-2133 in Research
Recently, The FRONTIER 2 long-term extension study, presented at the 2024 American Academy of Dermatology Annual Meeting in San Diego, showed that the JNJ-2113 treatment maintains its effectiveness from Week 16 through Week 52, with safety results aligning with those found in the earlier FRONTIER 1 study. Johnson & Johnson revealed initial outcomes from the FRONTIER 2 study, an extension of the Phase 2b FRONTIER 1 trial, which explores the use of JNJ-2113. This unique, targeted oral peptide aims to inhibit the IL-23 receptor, a key player in the activation of pathogenic T-cells associated with moderate-to-severe plaque psoriasis and other IL-23-mediated conditions in dermatology, rheumatology, and gastroenterology.
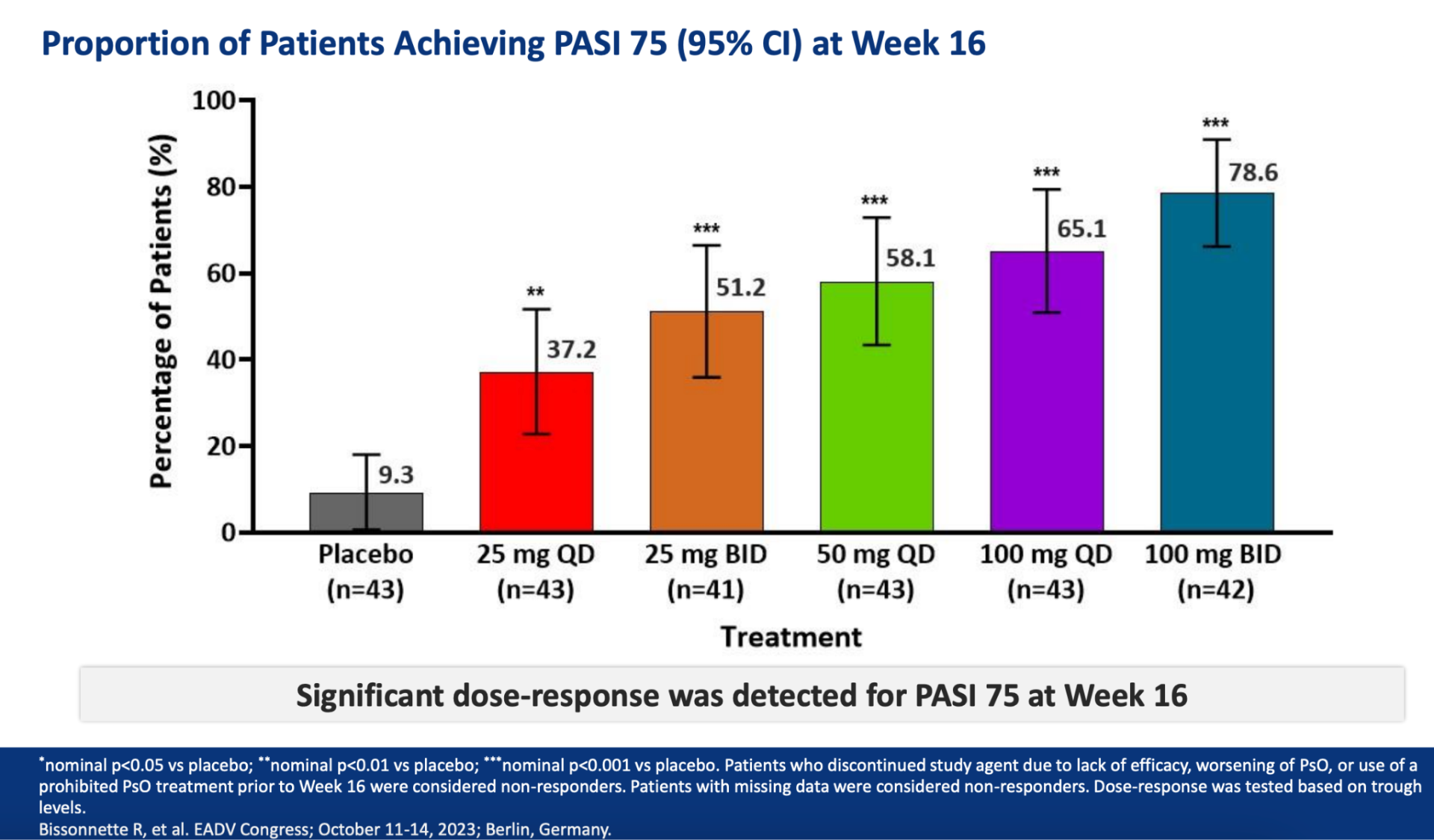
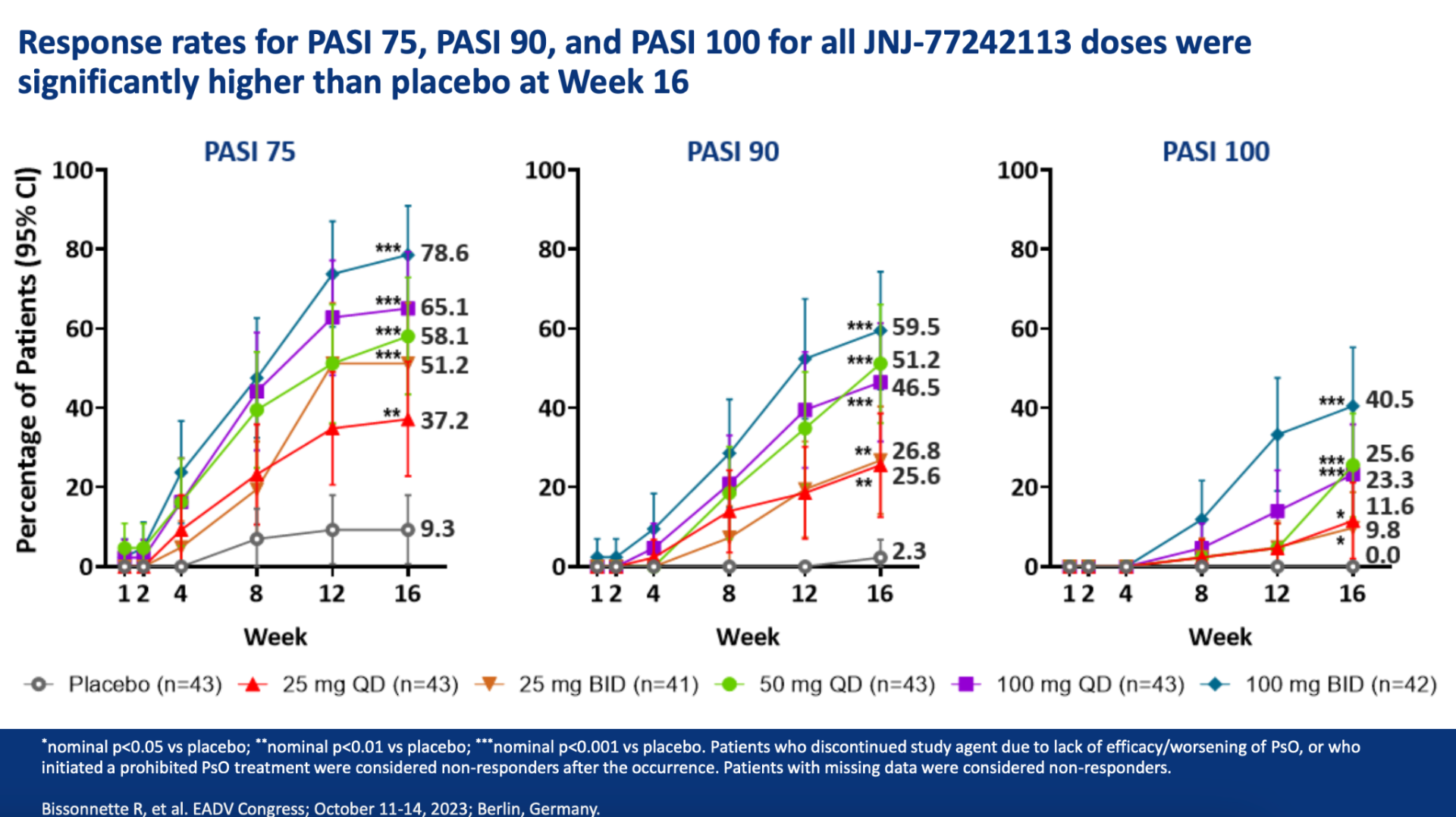
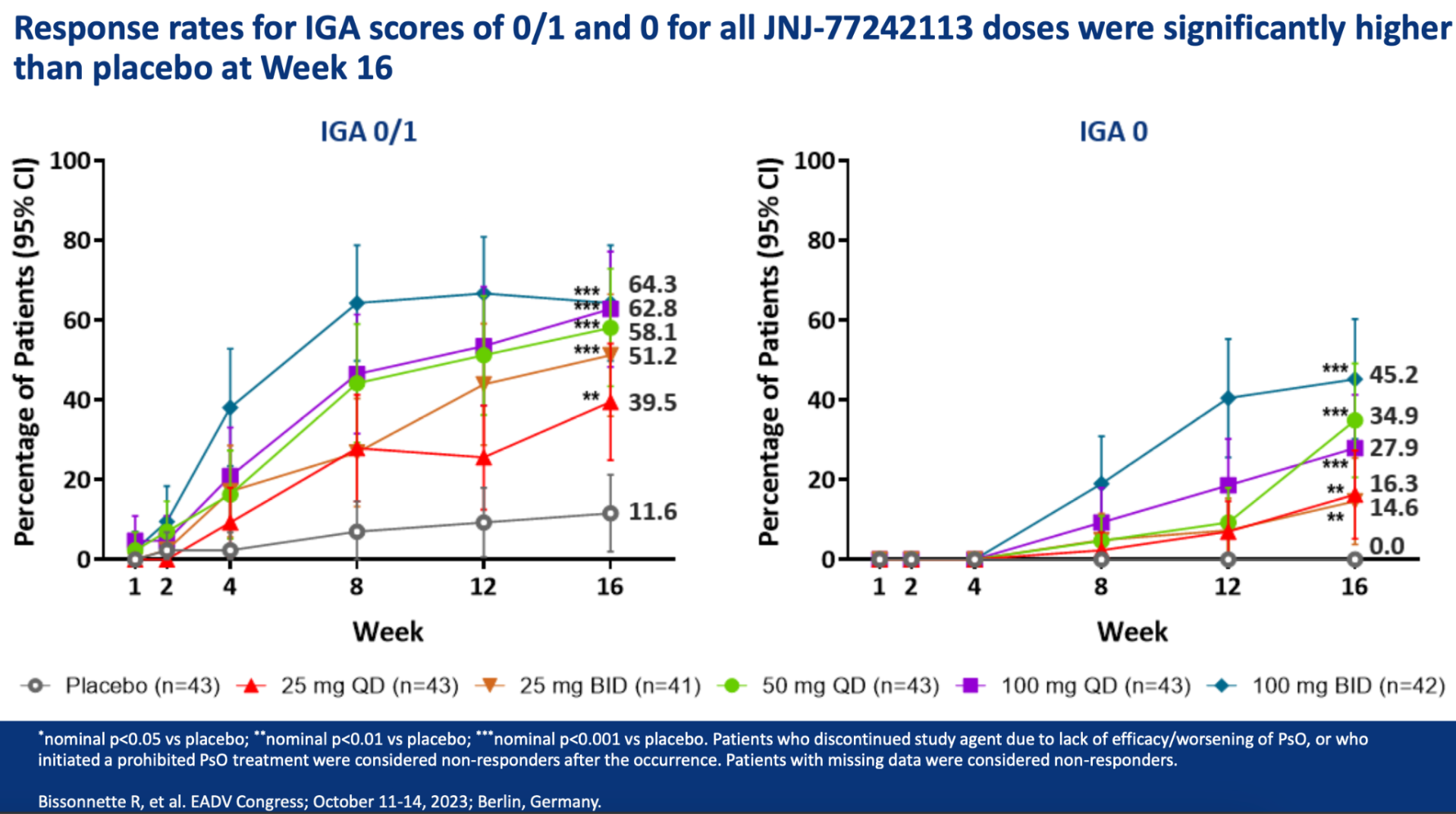
Throughout the year-long FRONTIER 2 study, adults with moderate-to-severe plaque psoriasis experienced significant and sustained skin clearance. Across five different dosage groups of JNJ-2113, measured by the Psoriasis Area and Severity Index, the effectiveness of the treatment was consistently upheld from Week 16 to Week 52. Notably, the 100 mg twice-daily group showed the highest skin clearance rates, with a 78.6% response at 16 weeks and a 76.2% response at 52 weeks. Similarly, for key secondary goals like PASI 90, PASI 100, and Investigator’s Global Assessment scores, effectiveness was maintained up to Week 52 for all dosage groups.
The safety profile observed from Week 16 to Week 52 in the FRONTIER 2 study was consistent with the findings from the initial FRONTIER 1 study. In the different JNJ-2113 treatment groups, 58.6% of participants encountered adverse events (AEs), with no increase in AEs related to higher doses, including those affecting the gastrointestinal system. The most common AEs reported were nasopharyngitis (18.1%), upper respiratory infections (9.7%), and COVID-19 (5.3%). Serious AEs were rare, occurring in only 4% of participants treated with JNJ-2113 by Week 52, and none were deemed related to the treatment.
Laura Ferris, M.D., Ph.D., a Dermatology Professor at the University of Pittsburgh, remarked, “The FRONTIER 2 study data confirms that the skin improvement noted at 16 weeks, such as PASI 75, and the more stringent PASI 90 and 100 outcomes, were sustained at 52 weeks without any new safety concerns in all JNJ-2113 treatment scenarios. These outcomes indicate that JNJ-2113 could fulfill the significant unmet need for a new, long-lasting, and easy-to-use oral treatment for individuals with moderate-to-severe plaque psoriasis.”
In the continuation phase, FRONTIER 2, participants from the FRONTIER 1 study who initially received JNJ-2113 continued on the same dose. Those who were in the placebo group for the first part of the study were administered JNJ-2113 100 mg once daily in the subsequent study. The results of the Phase 2b FRONTIER 1 trial, which assessed JNJ-2113 in adults with moderate-to-severe plaque psoriasis, have been recently published in The New England Journal of Medicine.
The crucial ICONIC Phase 3 clinical trial series is currently being conducted to assess the safety and effectiveness of JNJ-2113 in adults experiencing moderate-to-severe plaque psoriasis, focusing on studies such as ICONIC-LEADe and ICONIC-TOTAL, following an agreement between Protagonist Therapeutics, Inc. and Janssen Biotech, Inc. Additionally, the program has launched other Phase 3 trials, namely ICONIC-ADVANCE 1 and ICONIC-ADVANCE 2, to compare the safety and effectiveness of JNJ-2113 with both a placebo and deucravacitinib.
Results from the FRONTIER 1 and FRONTIER 2 trials highlight the potential usefulness of JNJ-2113 for a variety of IL-23-related conditions. Consequently, Johnson & Johnson has started the Phase 2b ANTHEM-UC study, which aims to assess the safety and efficacy of JNJ-2113 against a placebo in individuals with moderate to severe active ulcerative colitis.
Lloyd Miller, M.D., Ph.D., Vice President and Immunodermatology Disease Area Leader at Johnson & Johnson, expressed, “Our dedication to innovation in the field of Immunodermatology is in line with our broader goal to introduce new therapeutic solutions to achieve disease remission in individuals facing immune-mediated diseases. The encouraging findings after one year from the FRONTIER 2 study demonstrate our commitment to pioneering within the IL-23 pathway, aiming to discover novel and distinct treatment avenues for psoriasis patients.”
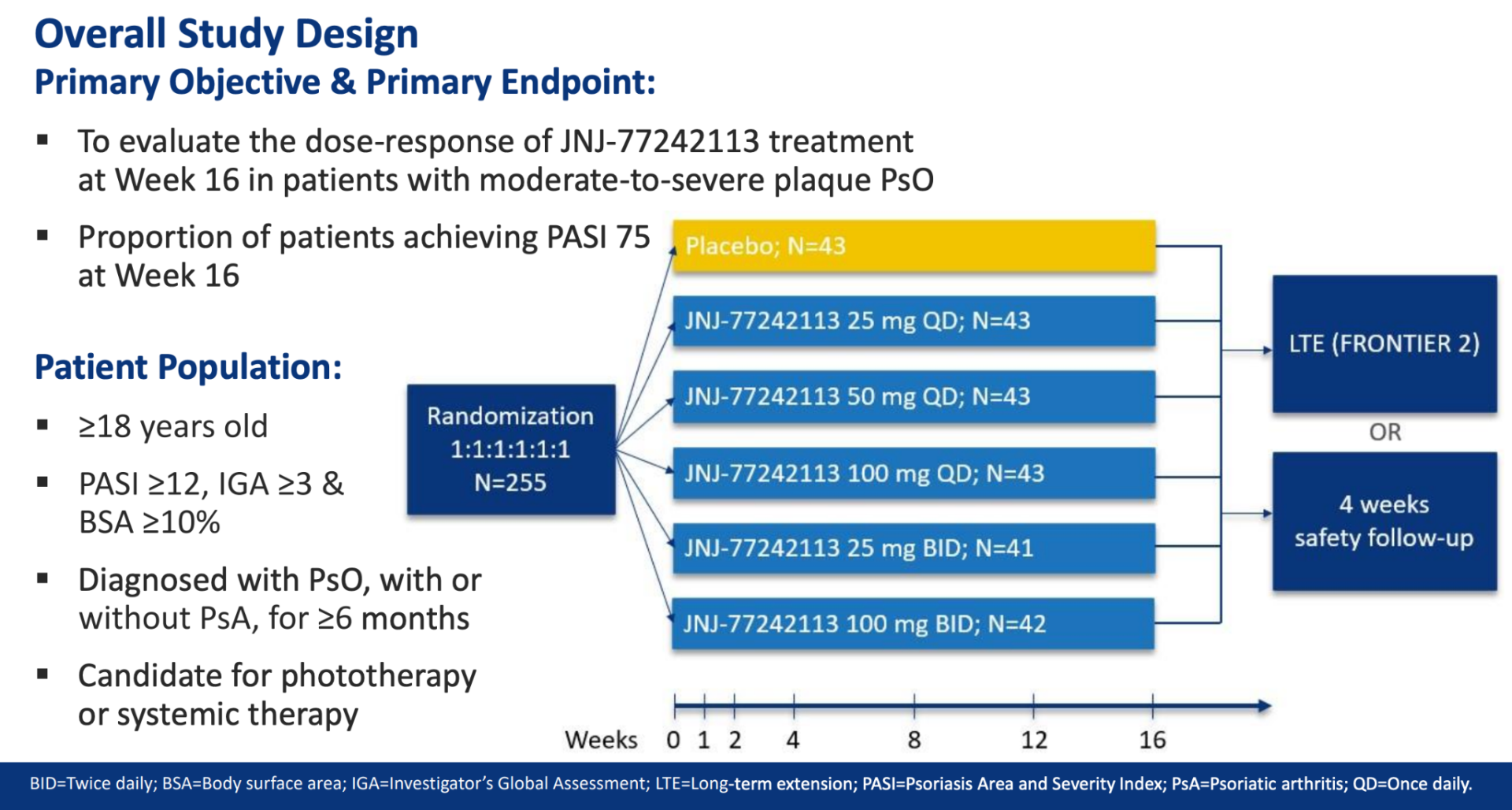
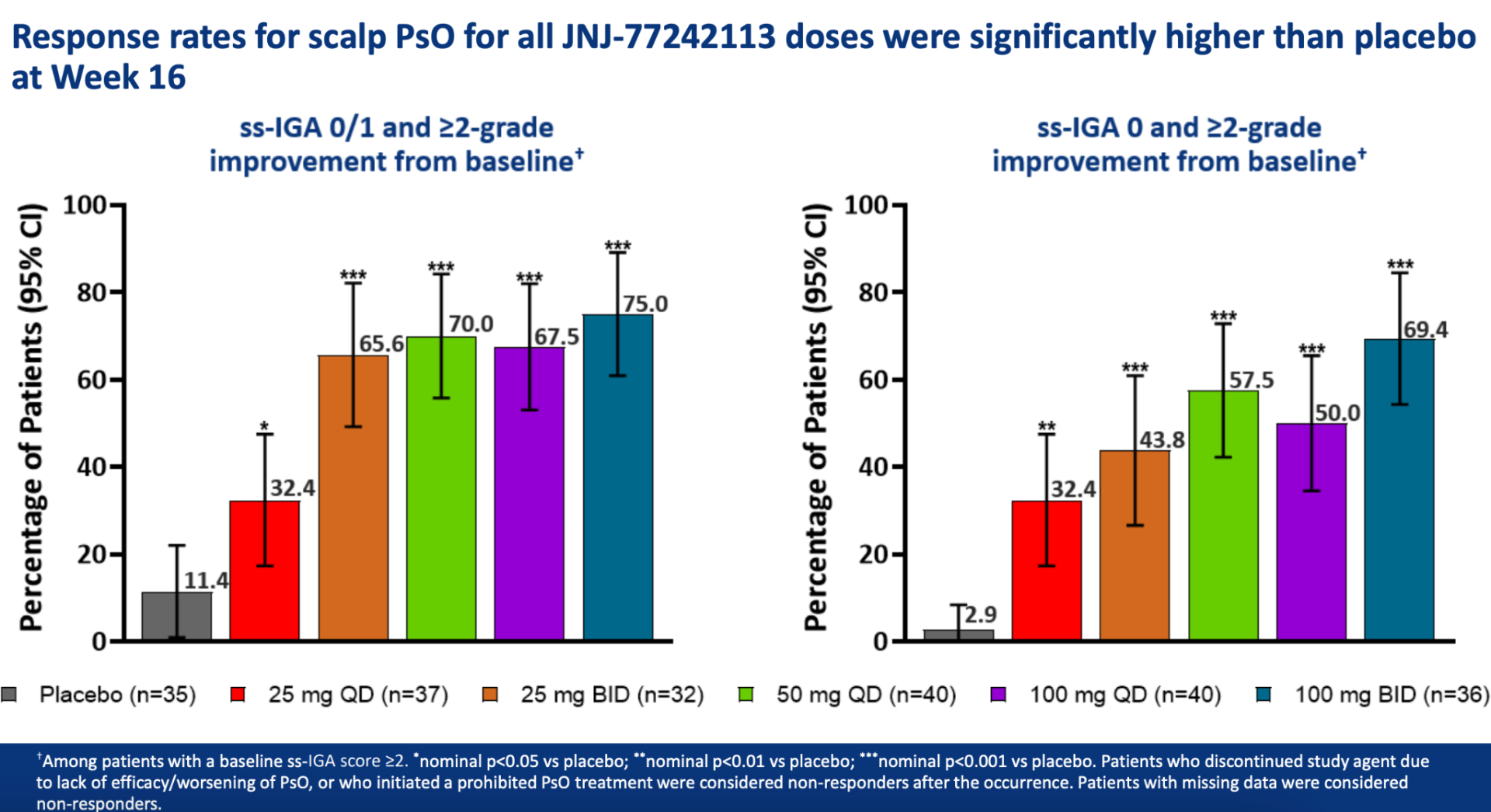
You can view the study design and results from A Phase 2, Randomized, Placebo-Controlled, Dose-Ranging Study of Oral JNJ-77242113 for the Treatment of Moderate-to-Severe Plaque Psoriasis: Efficacy of Overall and Scalp Psoriasis Responses From FRONTIER 1 below.