Uncategorized
Part 2: “Cellular Senescence: What Can Be Done About It?”
Brief overview/Summary
Cellular senescence is a natural process with both protective and harmful effects. Throughout life, senescence helps prevent tumor formation and mitigate tissue damage. However, as individuals age, senescent cells accumulate in tissues, potentially contributing to various age-related diseases. Recent research has uncovered the molecular mechanisms that support the survival of senescent cells and regulate their immune clearance. These findings provide a foundation for developing new therapeutic approaches to target senescent cells while highlighting the importance of understanding the limitations, efficacy, safety, and potential risks of current strategies for senescent cell elimination.
This article explores existing methods for targeting senescent cells and the challenges in advancing these strategies into safe and effective therapies. Successfully addressing these challenges could revolutionize treatments for age-related diseases and transform the way we approach health management during aging.
Introduction
The global population is aging rapidly, presenting both opportunities and significant challenges, particularly as advanced age increases susceptibility to chronic diseases. Aging is the leading risk factor for many of the world’s most prevalent conditions, including cardiovascular diseases, cancer, and neurodegenerative disorders. However, aging is not uniform—some individuals maintain better health and functionality than others at the same chronological age, experiencing longer periods of well-being. This underscores the importance of understanding the shared cellular and molecular pathways that drive age-related multimorbidity’s. Targeting these pathways may offer more effective treatments than addressing each disease in isolation. Recent discoveries have shed light on the biological mechanisms underlying aging, with one key factor being the accumulation of senescent cells in tissues.
Cellular senescence is a largely irreversible arrest of the cell cycle in normal, proliferating cells, triggered by various stressors such as replicative exhaustion, oncogene activation, DNA damage, and cell-cell fusion. This response, mediated by the DNA damage response pathway, serves as a protective mechanism to prevent the propagation of damaged cells. Cellular senescence plays crucial roles in tumor suppression, limiting tissue damage, and aiding wound healing. However, despite these protective effects, the persistent presence of senescent cells can have detrimental consequences.
Senescent cells secrete a complex mix of proinflammatory factors, which, while aiding their removal by the immune system, can contribute to chronic inflammation when these cells accumulate. Evidence from studies on animal models suggests that components of the immune system, such as NK cells, T cells, and macrophages, play a role in regulating senescent cell presence in tissues. However, the effectiveness of this immune-mediated clearance varies depending on the tissue and pathological conditions, and the mechanisms governing the balance of senescent cells remain incompletely understood.
Senescent cells secrete a complex mix of proinflammatory factors, which, while aiding their removal by the immune system, can contribute to chronic inflammation when these cells accumulate. Evidence from studies on animal models suggests that components of the immune system, such as NK cells, T cells, and macrophages, play a role in regulating senescent cell presence in tissues. However, the effectiveness of this immune-mediated clearance varies depending on the tissue and pathological conditions, and the mechanisms governing the balance of senescent cells remain incompletely understood.
In advanced age, senescent cells accumulate in tissues and promote a state of chronic, low-grade inflammation—termed “inflammaging”—through continuous secretion of proinflammatory cytokines. This phenomenon is closely associated with most age-related diseases. Senescent cells are particularly prevalent in areas affected by age-related pathologies, and mounting evidence from mouse models indicates that these cells play a causal role in conditions such as atherosclerosis, idiopathic pulmonary fibrosis, osteoarthritis, bone loss, and hepatic steatosis. Notably, genetic strategies to enhance the clearance of senescent cells expressing the p16 marker in mice have been shown to delay the onset of age-related tissue deterioration and extend median lifespan.
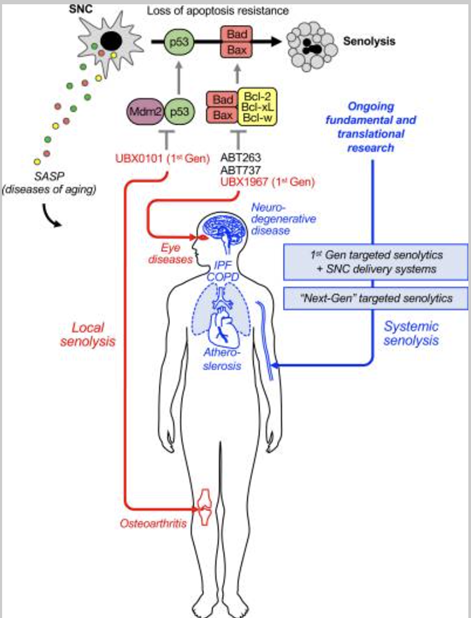
Therapeutic Strategies for Targeting Senescent Cells
Interest in targeting senescent cells therapeutically is rapidly growing, with several promising strategies under development. These approaches focus on either clearing senescent cells or mitigating their proinflammatory effects. A key area of research involves the discovery of pharmacological agents, known as “senolytics,” which can selectively induce the death of senescent cells.
These efforts leverage the unique molecular characteristics of senescent cells, particularly their ability to accumulate with age and their distinct biological features compared to other cell types. One notable hallmark of senescent cells is their resistance to apoptosis. Unlike normal cells, senescent cells are shielded from both intrinsic and extrinsic proapoptotic signals, enabling them to persist and influence various biological processes even under stress.
By targeting these apoptotic pathways specifically in senescent cells, senolytic therapies can induce their selective elimination, effectively preventing the harmful effects associated with their accumulation. This pharmacological approach differs in several respects from genetic strategies but offers a compelling method to address the detrimental impact of senescent cells on aging and age-related diseases.



Three principal approaches could be implemented to block deleterious effects of senescent cells. (A) The leading options are strategies that induce apoptosis selectively in senescent cells with the use of senolytic drugs, which block pro-survival pathways. (B) The second type of strategy is to potentiate an immune response against senescent cells in a way that would lead to their clearance from tissues. (C) A third strategy proposes specific blockade of SASP components by targeting their upstream regulators and effectors. ADCC, antibody-dependent cell-mediated cytotoxicity; DDR, DNA damage response; GOS, galacto-oligosaccharides; ITAM, immunoreceptor tyrosine-based activation motif.
Blocking Survival Pathways
Several pro-survival pathways have been identified as potential targets for the directed elimination of senescent cells, including the BCL-2 protein family, the p53/p21 axis, PI3K/AKT, receptor tyrosine kinases, and the HIF-1α and HSP90 proteins. Among these, the BCL-2 protein family has garnered significant attention due to its role in regulating apoptosis and autophagy, making it a particularly attractive drug target. During senescence, antiapoptotic proteins such as BCL-2, BCL-W, and BCL-XL are upregulated in various cell types, and silencing these proteins triggers programmed cell death.
Pharmacological inhibitors of the BCL-2 family have been extensively studied in cancer therapy and have shown promise as senolytics. ABT-737, a BH3 mimetic, disrupts the interaction between antiapoptotic BCL-2 family members and proapoptotic proteins, allowing senescent cells to undergo apoptosis. In mouse models, ABT-737 effectively clears senescent cells induced by DNA damage or p14ARF activation, even promoting hair follicle stem cell proliferation and hair growth. A next-generation analog, ABT-263 (navitoclax), has demonstrated the ability to eliminate senescent muscle and hematopoietic stem cells in aged and irradiated mice, rejuvenating these populations. It also removes senescent foam cell macrophages from atherosclerotic plaques, halting disease progression.
Despite these benefits, general inhibitors of BCL-2, BCL-W, and BCL-XL, such as ABT-737 and ABT-263, cause hematological toxicities, including neutropenia and thrombocytopenia. These side effects highlight the need for more selective inhibitors. For example, BCL-XL-specific inhibitors like A1331852 and A1155463 may reduce toxicity in non-senescent cells while still inducing apoptosis in senescent cholangiocytes and fibroblasts, as demonstrated in a mouse model of biliary liver fibrosis. However, platelet survival depends on BCL-XL, so transient thrombocytopenia remains a concern, and these inhibitors may be less effective against senescent cells expressing higher levels of BCL-2 or BCL-W.
Localized administration of BCL-2 family inhibitors could mitigate systemic side effects. For instance, intra-articular injection of UBX0101, a BCL-2-targeting compound, successfully cleared senescent cells from articular cartilage and synovium, reducing osteoarthritis symptoms in aged mice. While targeting the BCL-2 family represents a validated approach for senescent cell clearance, its clinical utility may be limited by associated toxicities, underscoring the need for more precise and localized therapeutic strategies.
Senolytic Combinations
A potential strategy to mitigate the cytotoxic side effects of BCL-2 protein family inhibitors is to combine low doses of these inhibitors with compounds that target other senescence-associated pro-survival pathways. Notably, the first senolytic discovered was not a single compound but a combination of the pan-tyrosine kinase inhibitor dasatinib and the naturally occurring flavonoid quercetin. Together, these compounds were shown to selectively eliminate senescent primary preadipocytes and senescent human umbilical vein endothelial cells (HUVECs) in tissue culture and reduce senescent cell signatures across several pathological conditions. In animal models, dasatinib/quercetin reduced aortic calcification and osteogenic signaling in aged and hypercholesterolemic mice and improved lung function in mice with bleomycin-induced lung fibrosis. However, the combination failed to address the fibrotic components of these conditions, suggesting its greater utility as a preventative intervention in early disease stages. Supporting this, dasatinib/quercetin has been shown to prevent age-related bone loss and reduce hepatic steatosis, a precursor to nonalcoholic fatty liver disease.
Both dasatinib and quercetin have been tested in clinical settings, making this combination a promising candidate for senolytic therapy. However, several challenges may limit its broader application. For one, both compounds target numerous biological pathways, and the mechanisms underlying their senolytic effects are not fully understood. Furthermore, the combination’s effects likely arise from alterations in multiple pathways across various tissues and organs, raising concerns about potential adverse effects with long-term use. Nevertheless, short-term administration of the combination has proven sufficient to eliminate senescent cells and confer therapeutic benefits in preclinical studies. Since continuous presence of the compounds may not be necessary, the risk of off-target complications could be minimized.
Beyond quercetin, other natural compounds such as fisetin and piperlongumine have also demonstrated senolytic potential. These molecules are believed to target multiple molecular pathways to eliminate senescent cells. However, their unclear mechanisms of action and poor pharmacokinetic profiles make it unlikely that they will be developed into clinical therapies.
Targeting Senescence Specific Pathways
Recent studies have identified the p53/p21 axis as a promising target for developing novel senolytics. p53, a key tumor suppressor gene and the most frequently mutated in human cancers, is a central regulator of the senescence program. Stabilized in response to DNA damage, p53 accumulates and exerts transcriptional control over numerous processes, acting as a critical decision point between transient cell cycle arrest, senescence, and apoptosis. Disrupting the interaction between p53 and the transcription factor FOXO4 induces the release of p53 from the nucleus, triggering cell-intrinsic apoptosis. Administration of a modified peptide that interferes with the p53-FOXO4 interaction reversed liver chemotoxicity from doxorubicin in mice, while improving fitness, hair density, and kidney function in progeroid and naturally aged mice. Beyond the p53-FOXO4 interaction, p21 (CDKN1A), a primary transcriptional target of p53, also plays a vital role in senescent cell survival and retention. Following DNA damage, p21 prevents NF-κB–mediated JNK activation, caspase-3 cleavage, and subsequent apoptosis. In mouse models, p21 knockout reduced senescent cells in fibrotic livers and alleviated fibrosis. While indirect modulation of the p53/p21 axis, such as controlling ROS levels, is possible, no drugs currently induce apoptosis in senescent cells by directly inhibiting p53 or p21. Further research is essential to realize the therapeutic potential of this pathway. The FOXO4 peptide represents a promising starting point for senolytic development, and peptide-based or mimetic approaches could form the basis for future treatments.
The discovery of molecular pathways supporting senescent cell viability has been crucial for senolytic development. In addition to targeting specific pathways, high-throughput methods have identified novel targets, including the HSP90 protein family. HSP90, a group of molecular chaperones, promotes cell survival by stabilizing signaling proteins like AKT and ERK, which are upregulated during senescence. Disrupting the HSP90-AKT interaction inhibits the PI3K/AKT pathway, selectively killing senescent cells from various origins. In vivo, treatment with the HSP90 inhibitor 17-DMAG in progeroid mice reduced senescence markers and extended health span. These findings suggest HSP90 inhibitors could play a critical role in combinatorial senolytic therapies, paving the way for more effective interventions.
Directed Elimination of Senescence Cells
Targeting senescence-specific pathways is a promising strategy for developing senolytics, but other unique features of senescent cells can also be exploited for their elimination. One such feature is the increased activity of senescence-associated β-galactosidase (SA β-gal). Leveraging this, researchers have designed a delivery system using mesoporous silica nanoparticles coated with galacto-oligosaccharides. These nanoparticles remain inactive in non-senescent cells but are activated in senescent cells, where the coating is digested, allowing the nanoparticle contents to be released. For instance, cytotoxic drugs encapsulated in these coated nanoparticles could be selectively released into the cytoplasm of senescent cells, inducing apoptosis.
However, SA β-gal activity is not an exclusive marker of senescent cells, as certain other cell types, such as macrophages, can exhibit elevated β-galactosidase activity under specific conditions). To enhance selectivity, these nanoparticles could be tethered to antibodies targeting known surface markers unique to senescent cells. While senolytic strategies based on SA β-gal activity are conceptually promising, they have yet to be tested in vivo. Further studies are required to evaluate their efficacy and safety in treating senescence-associated conditions.
Immune Mediated Interventions
Beyond directly eliminating senescent cells based on their intrinsic properties, therapeutic interventions can leverage the immune system. Senescent cells are naturally immunogenic and subject to immune surveillance mechanisms. In a hepatocellular carcinoma model, p53 restoration induces senescence and triggers an innate immune response involving leukocytes, neutrophils, macrophages, and NK cells. Similarly, in a liver fibrosis model, senescent hepatic stellate cells promote their elimination by NK cells. Senescent cells often upregulate NKG2D, an immune recognition molecule absent on normal cells, making it a promising target for immunotherapy approaches. However, senescence surveillance mechanisms vary by context. For instance, senescence induced by NRASG12V expression in hepatocytes results in a mixed immune response, with CD4+ T cells and monocytes/macrophages cooperating to clear senescent cells. The accumulation of senescent cells in aged tissues may result partly from immune decline with age. Enhancing immune function could support the clearance of these cells. Supporting this, the immune stimulator polyI:C facilitates NK cell–mediated clearance of senescent cells in fibrotic livers. However, such strong immune stimulators may overwhelm the already chronically activated aging immune system, necessitating more precise modulators.
Adopting immunotherapy strategies from cancer treatment could aid these efforts. For example, blocking the immune checkpoint programmed death 1 (PD-1), a powerful cancer therapy, also alleviates symptoms in a murine model of Alzheimer’s disease. Additionally, tools like chimeric antigen receptor (CAR) T cells could redirect immune responses to target senescent cells. However, a major challenge in immune-based approaches is the limited availability of highly specific senescence markers. Recent studies have identified promising candidates. Senescent fibroblasts were found to express an oxidized form of membrane-bound vimentin on their surface, and mass spectrometry identified dipeptidyl peptidase 4 (DPP4) as a cell surface marker for senescent fibroblasts. DPP4’s selective expression allows NK cells to target senescent cells via antibody-dependent cell-mediated cytotoxicity (ADCC). However, senescent cells are partially protected from NK cell–mediated killing due to upregulation of decoy receptor 2 (DcR2), which inhibits death receptor activation by TNF-related apoptosis-inducing ligand (TRAIL), confining cytotoxicity to perforin- and granzyme-mediated pathways. Strategies to block DcR2 could enhance senescent cell vulnerability to natural immune clearance and immunotherapy.
Strategies Aimed to Limit Detrimental Roles of Senescent Cells
While eliminating senescent cells is a promising strategy, it may not be suitable for all conditions where senescent cells are present. In such cases, targeting the harmful components of senescence could serve as an alternative approach. Senescent cells exhibit a consistent proinflammatory secretory profile, known as the senescence-associated secretory phenotype (SASP), which is conserved across different senescent states and cell types. The SASP consists of inflammatory chemokines, cytokines, growth factors, and matrix-remodeling proteases. While the SASP plays a role in tumor suppression by enforcing cell cycle arrest in damaged cells and recruiting immune cells for clearance, it is also implicated in disrupting tissue homeostasis and driving age-related pathologies. Several signaling pathways, including the NF-κB and C/EBPβ pathways, regulate the SASP and can be modulated by various FDA-approved drugs. For example, IL-1α signaling upstream of NF-κB can be inhibited with neutralizing antibodies against IL-1α or its receptor, reducing NF-κB activity. mTOR inhibitors like rapamycin can suppress SASP by lowering IL-1α expression, mitigating senescent fibroblasts’ ability to promote tumor growth in mice. Metformin, widely used for type 2 diabetes, blocks NF-κB nuclear translocation, limiting inflammation, alleviating age-related pathologies, and extending lifespan in mice. Ongoing studies explore its anti-aging potential in non-diabetic populations. C/EBPβ activity, induced by JAK/STAT signaling, can be reduced with JAK1/2 inhibitors like ruxolitinib, improving systemic inflammation and fitness in aged mice. These findings suggest that existing drugs may dampen the SASP’s effects in vivo, potentially mitigating the negative impact of senescent cells.
Restricting inflammation to limit the harmful effects of senescent cells shows promise, but prolonged use of potent anti-inflammatory drugs may cause side effects. This limitation could hinder long-term treatment necessary to retain senescence’s beneficial effects. Recent studies on epigenetic regulators such as MLL1, BRD4, and HMGB2 may provide a solution. These proteins reshape the epigenetic landscape of senescent cells, influencing SASP expression. Knocking down these chromatin modifiers sustainably suppresses inflammation without affecting cell cycle arrest. Small-molecule modulators, including bromodomain and extra-terminal motif (BET) inhibitors, could be explored further for their potential in this area. Targeting specific SASP components may also provide a safer approach. Well-defined SASP cytokines like IL-6 and IL-8 or matrix-remodeling proteases like ADAM17 represent viable targets. Neutralizing antibodies, developed from commercially available monoclonal antibodies, could block these molecules effectively. For instance, IL-6 or its receptor can be targeted with approved drugs like siltuximab or tocilizumab, which block the cytokine or its receptor, respectively. However, these agents were not initially designed for senescence-related applications, and further research is needed to assess their effects on SASP and aging before therapeutic use as senescence modifiers.
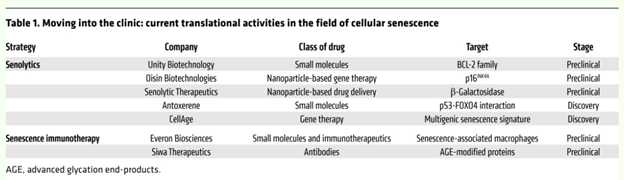
Moving Toward Clinical Trials
Successful translation of strategies aimed at targeting senescent cells into the clinic could have a vast impact on the treatment of a variety of diseases at old age and potentially revolutionize our view of aging. Several technologies aiming at senescent cells are now taking their first steps in the challenging, time-consuming, and expensive journey of drug development. In order to maximize their chances of becoming common treatments, critical issues of efficacy, safety, and tolerability should be considered during the early stages of development.
Sources:
Product available for research use only:

Posted in:FOXO4-DRICellular Senesence
