Tirzepatide 5mg
$195.00
Buy Tirzepatide 5mg online. A clinically proven GIP & GLP-1 receptor agonist for type 2 diabetes control and significant weight loss. Once-weekly. Doctor-trusted.
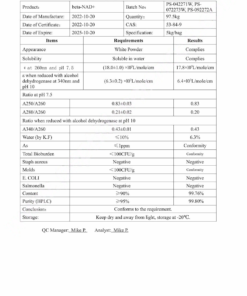
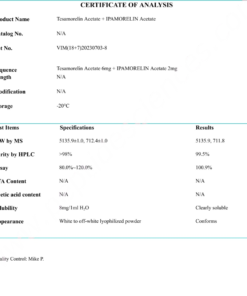
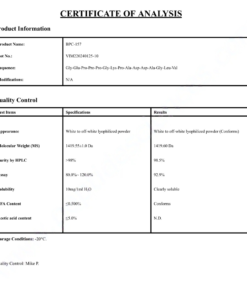
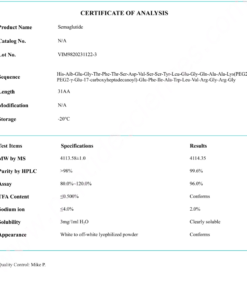
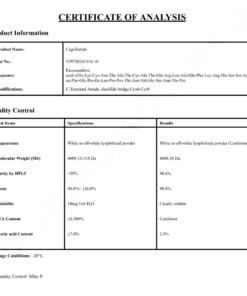
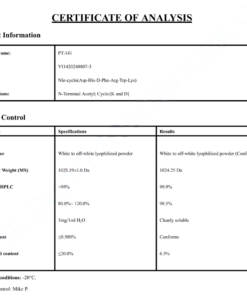
COA Tirzepatide 5mg Certificate
Tirzepatide 5mg — The Next-Generation Solution for Type 2 Diabetes and Weight Management
Tirzepatide 5mg is a groundbreaking injectable medication that offers a dual-action mechanism uniquely designed to improve blood sugar control and support meaningful weight loss. As the first-in-class glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor agonist, Tirzepatide 5mg represents a major advancement in the treatment of type 2 diabetes mellitus in adults. Whether prescribed for glycemic control or as part of a comprehensive weight management plan, this medication delivers clinically significant results backed by extensive research.
HPLC Tirzepatide 5mg Certificate
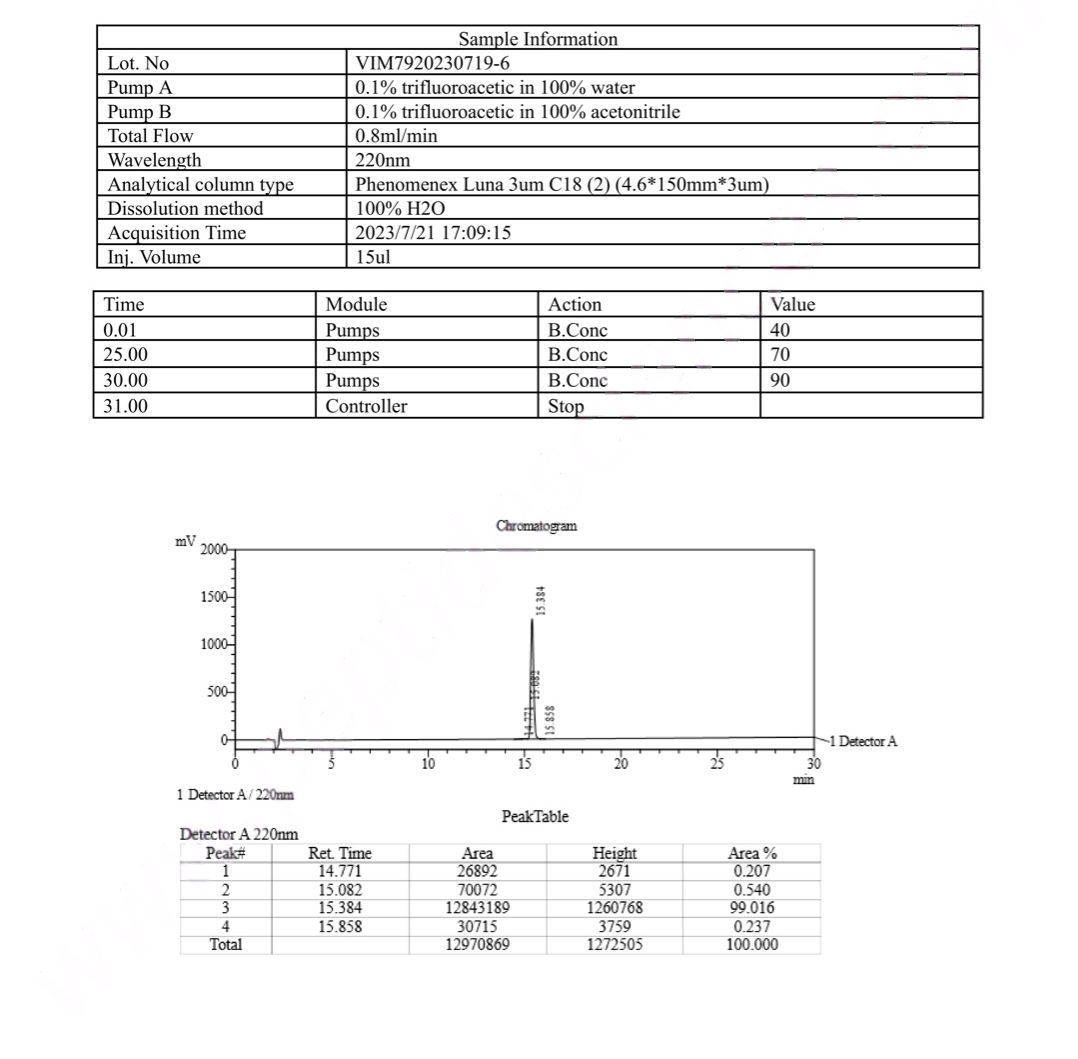
MS Tirzepatide 5mg Certificate
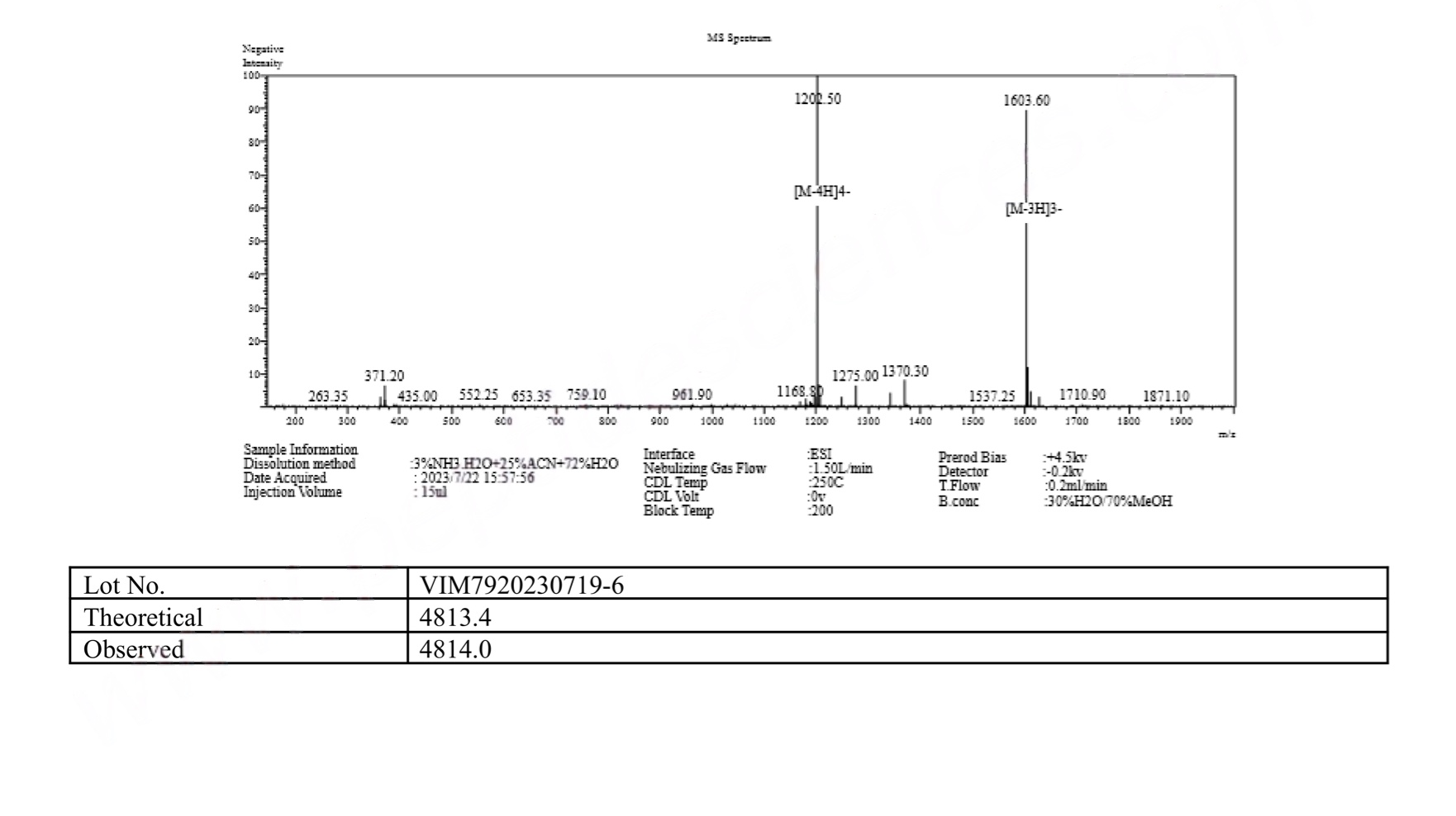
What Is Tirzepatide 5mg?
Tirzepatide 5mg is a once-weekly subcutaneous injection developed by Eli Lilly and Company and marketed under the brand name Mounjaro for type 2 diabetes. The 5mg dose is a maintenance and titration dose, typically reached after a patient starts on a lower 2.5mg dose during the initial weeks of treatment. Tirzepatide works by simultaneously activating both GIP and GLP-1 receptors in the body, two key hormones involved in regulating blood sugar and appetite. This dual receptor activation sets Tirzepatide apart from older GLP-1-only therapies, delivering superior outcomes in both glycemic control and body weight reduction.
How Tirzepatide 5mg Works: Dual Hormone Action
The effectiveness of Tirzepatide 5mg lies in its innovative dual mechanism of action:
GIP Receptor Activation
GIP (glucose-dependent insulinotropic polypeptide) is an incretin hormone that stimulates insulin secretion in response to food intake. By activating GIP receptors, Tirzepatide enhances the body’s natural insulin response after meals, helping to lower post-meal blood glucose spikes without causing hypoglycemia when blood sugar is already within a safe range.
GLP-1 Receptor Activation
GLP-1 (glucagon-like peptide-1) activation reduces glucagon secretion, slows gastric emptying, and promotes a feeling of fullness (satiety). Together, these effects lead to reduced appetite and caloric intake, which supports sustained weight loss alongside improved blood sugar management.
Key Benefits of Tirzepatide 5mg
Clinically proven advantages include:
- Significant HbA1c reduction — clinical trials show reductions of up to 2.0% or more
- Meaningful weight loss — patients lost an average of 15–20% of body weight in SURPASS trials
- Once-weekly dosing — convenient self-injection using a pre-filled pen device
- Cardiovascular benefits — associated with improvements in blood pressure and lipid profiles
- Low hypoglycemia risk — glucose-dependent mechanism reduces the chance of low blood sugar episodes
- Durable results — sustained glycemic control over long-term treatment
Tirzepatide 5mg Dosage and Administration
Tirzepatide is administered as a subcutaneous injection once per week, on the same day each week, with or without meals. The recommended starting dose is 2.5mg per week for the first four weeks to minimize gastrointestinal side effects. After four weeks, the dose is increased to 5mg once weekly. Based on individual patient response and tolerability, the dose may be further titrated in 2.5mg increments every four weeks, up to a maximum of 15mg per week. Injection sites include the abdomen, thigh, or upper arm, and should be rotated with each dose.
Who Should Use Tirzepatide 5mg?
Tirzepatide 5mg is indicated for adults diagnosed with type 2 diabetes mellitus as an adjunct to diet and exercise. It is particularly beneficial for patients who need both glycemic control and weight reduction. It is not indicated for patients with type 1 diabetes or as a treatment for diabetic ketoacidosis. Always consult a qualified healthcare provider before starting or adjusting any diabetes treatment.
Common Side Effects and Safety Considerations
As with all medications, Tirzepatide 5mg may cause side effects. The most commonly reported are gastrointestinal in nature and tend to be mild to moderate, especially during the initial dose escalation period.
Most Commonly Reported Side Effects
- Nausea
- Diarrhea
- Vomiting
- Constipation
- Decreased appetite
- Injection site reactions
Serious but less common risks include pancreatitis, gallbladder disease, and a possible risk of thyroid C-cell tumors based on animal studies. Tirzepatide carries a boxed warning regarding thyroid cancer risk and is contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or in patients with Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
Storage and Handling of Tirzepatide 5mg
Store Tirzepatide 5mg pens in the refrigerator at 2°C to 8°C (36°F to 46°F). Do not freeze. If needed, an unopened pen can be stored at room temperature below 30°C (86°F) for up to 21 days. Keep away from direct heat and light. Do not use the pen if it has been frozen or if the solution appears cloudy, discolored, or contains particles.
Frequently Asked Questions About Tirzepatide 5mg
Is Tirzepatide 5mg FDA-Approved?
Yes. The FDA approved Tirzepatide (Mounjaro) in May 2022 for the treatment of type 2 diabetes in adults. It has since received significant attention for its weight loss efficacy, with Zepbound (Tirzepatide for obesity) receiving separate FDA approval in 2023.
How Is Tirzepatide Different From Semaglutide (Ozempic/Wegovy)?
While Semaglutide is a GLP-1-only receptor agonist, Tirzepatide activates both GIP and GLP-1 receptors. Clinical head-to-head studies suggest Tirzepatide may produce greater reductions in HbA1c and body weight compared to Semaglutide, making it a superior option for many patients.
Be the first to review “Tirzepatide 5mg” Cancel reply
Related products
Peptide
Popular products
Popular products
Popular products
Popular products
Popular products
Popular products
Popular products




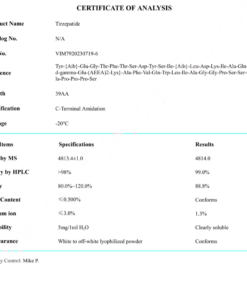










Reviews
There are no reviews yet.