Cagrilintide 10mg
$160.00
Cagrilintide 10mg is a once-weekly amylin analogue clinically studied for significant weight loss and metabolic health improvement. Explore dosage, mechanism of action, clinical benefits, and combination therapy insights for Cagrilintide 10mg.
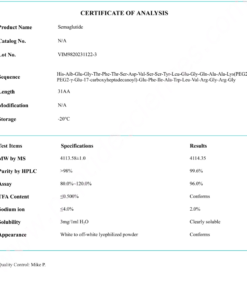
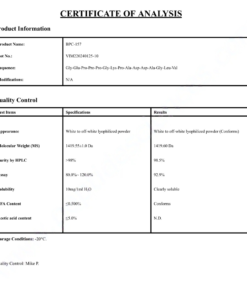
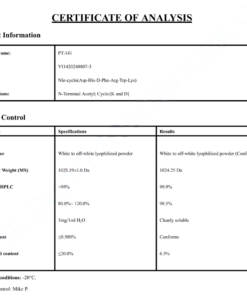
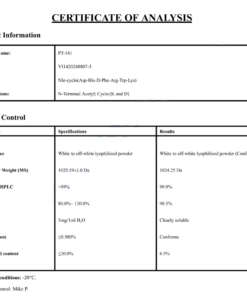
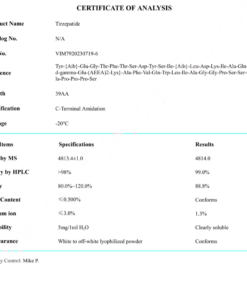
COA Cagrilintide 10mg Certificate
Cagrilintide 10mg — Advanced Amylin Receptor Agonist for Weight & Metabolic Management
Cagrilintide 10mg is a long-acting, acylated amylin analogue specifically engineered to support satiety signaling and metabolic regulation. Developed as a once-weekly subcutaneous injection, this investigational compound has attracted significant clinical attention for its potential role in obesity treatment and cardiometabolic health optimization. Its precise mechanism of action on amylin and calcitonin receptors sets it apart from conventional weight-loss peptides.
HPLC Cagrilintide 10mg Certificate
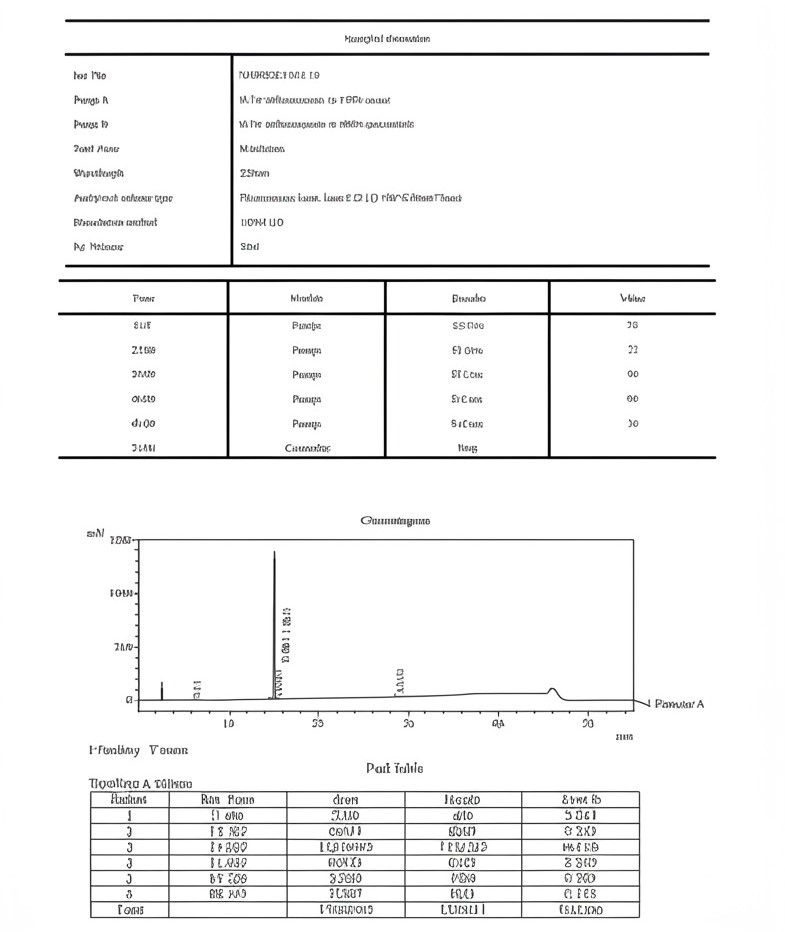
MS Cagrilintide 10mg Certificate
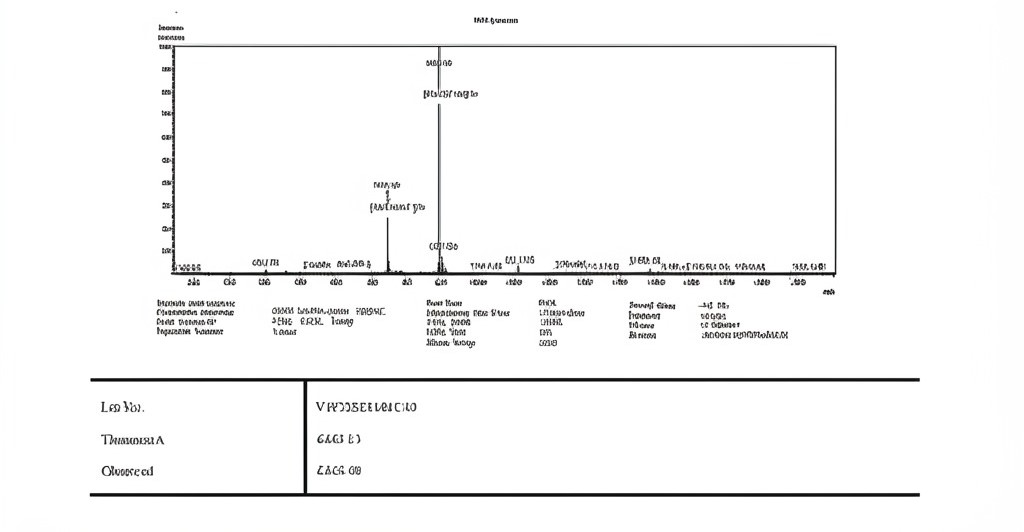
Product Name: Cagrilintide
Strength: 10mg
Drug Class: Long-Acting Amylin Analogue / Amylin Receptor Agonist
Route of Administration: Subcutaneous Injection (Once Weekly)
Research Status: Phase 3 Clinical Trials (CagriSema Program)
Target: Amylin Receptors (AMY1, AMY2, AMY3) & Calcitonin Receptors
How Cagrilintide 10mg Works: Mechanism of Action
Cagrilintide 10mg exerts its pharmacological effects by binding to amylin receptors in the central nervous system — primarily in the area postrema and hypothalamus — to modulate appetite, food intake, and gastric emptying. As a fatty-acid acylated amylin analogue, it is structurally engineered for extended half-life, enabling once-weekly dosing.
Key Pharmacological Actions of Cagrilintide
- Activates amylin and calcitonin receptors (AMY1-R, AMY2-R, AMY3-R)
- Reduces caloric intake by promoting central satiety signals
- Slows gastric emptying, prolonging the feeling of fullness after meals
- Modulates glucagon secretion to support postprandial glucose control
- Operates independently of GLP-1 receptors, enabling synergistic combination therapy
Clinical Benefits of Cagrilintide 10mg in Obesity & Metabolic Research
Cagrilintide 10mg has demonstrated compelling clinical outcomes in Phase 2 and Phase 3 research trials. As a standalone agent and in combination with semaglutide (the CagriSema regimen), it has shown statistically significant reductions in body weight and improvements across multiple cardiometabolic markers.
Weight Reduction Outcomes
In clinical studies, Cagrilintide 10mg administered once weekly produced meaningful percent reductions in total body weight. When combined with semaglutide 2.4mg in the REDEFINE program, participants achieved substantially greater weight loss compared to either agent alone — positioning the combination as a potential cornerstone of pharmacological obesity treatment.
Cardiometabolic Improvements
- Reduction in waist circumference and visceral fat mass
- Improved fasting blood glucose and insulin sensitivity markers
- Favorable effects on systolic blood pressure
- Reduction in triglycerides and improvement in lipid profiles
- Potential cardioprotective effects under ongoing investigation
Tolerability Profile
Cagrilintide 10mg has shown a manageable safety and tolerability profile in clinical settings. The most commonly reported adverse events are gastrointestinal in nature — including nausea, vomiting, and decreased appetite — which are typically mild-to-moderate and transient, particularly during the initial dose-escalation phase.
Cagrilintide 10mg Dosage, Administration & Dosing Schedule
Recommended Dosing Protocol
Cagrilintide is administered as a once-weekly subcutaneous injection. In clinical trials, dose escalation protocols were used to minimize gastrointestinal side effects, starting from lower doses and titrating up to the 10mg maintenance dose over several weeks. The 10mg dose represents the highest studied therapeutic dose in the CagriSema Phase 3 program.
Administration Guidelines
- Inject subcutaneously into the abdomen, thigh, or upper arm
- Rotate injection sites weekly to avoid lipodystrophy
- May be administered with or without food
- Store as directed — typically refrigerated at 2–8°C
- Use only under the supervision of a licensed healthcare provider
Cagrilintide in Combination Therapy: The CagriSema Approach
One of the most clinically significant developments surrounding Cagrilintide 10mg is its evaluation in combination with semaglutide — marketed as CagriSema. This dual-agonist strategy leverages complementary mechanisms: Cagrilintide’s amylin receptor activity pairs with semaglutide’s GLP-1 receptor agonism to produce additive or synergistic weight loss and metabolic benefits.
Why Combination Therapy Matters
Obesity is a multifactorial disease requiring multi-pathway intervention. By simultaneously targeting amylin and GLP-1 signaling, CagriSema addresses appetite, gastric emptying, glucagon suppression, and energy homeostasis through distinct yet complementary pathways. Emerging Phase 3 data suggests the combination may outperform existing approved monotherapies in percent body weight reduction.
Who May Benefit from Cagrilintide 10mg Research
Target Patient Populations in Clinical Studies
- Adults with obesity (BMI ≥ 30 kg/m²) with or without comorbidities
- Overweight adults (BMI ≥ 27 kg/m²) with weight-related health conditions
- Individuals with type 2 diabetes and excess body weight
- Patients for whom lifestyle interventions alone have been insufficient
Note: Cagrilintide 10mg is currently an investigational compound. It is not approved for general clinical use in most jurisdictions. Consult a qualified healthcare professional before considering any peptide-based therapy.
Frequently Asked Questions About Cagrilintide 10mg
Is Cagrilintide 10mg FDA Approved?
As of the latest available data, Cagrilintide 10mg has not received FDA or EMA approval as a standalone therapy. It is progressing through Phase 3 clinical trials. Regulatory submissions are anticipated following completion of the REDEFINE trial program.
What Is the Difference Between Cagrilintide and Semaglutide?
Cagrilintide acts on amylin and calcitonin receptors, while semaglutide is a GLP-1 receptor agonist. They target distinct biological pathways, which is precisely why their combination (CagriSema) is under investigation — offering broader metabolic coverage than either agent individually.
How Is Cagrilintide 10mg Supplied?
In clinical trial settings, Cagrilintide 10mg is supplied as a solution for subcutaneous injection in a prefilled pen or vial format. Exact commercial formulation details will be confirmed upon regulatory approval and market launch.
5 reviews for Cagrilintide 10mg
Add a review Cancel reply
Related products
Popular products
Popular products
Popular products
Popular products
Popular products
Popular products
Popular products
Popular products






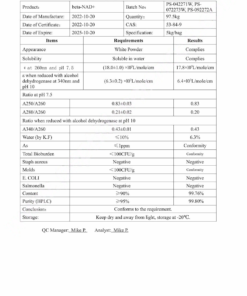







Jennifer Cole –
Helped me manage appetite and stay on track with my weight goals.
Ryan Bell –
Noticed reduced cravings and better energy throughout the day
Ashley Rivera –
Excellent support for weight management. Good value.
Brandon Murphy –
Cagrilintide is a game-changer. Works well with my routine
Kaitlyn Adams –
Pleased with the effects so far. Will monitor results.