Semaglutide
$115.00
Buy Semaglutide GLP-1 Analogue online — clinically proven for type 2 diabetes control and weight loss. Once-weekly injection. Prescribed by licensed doctors.
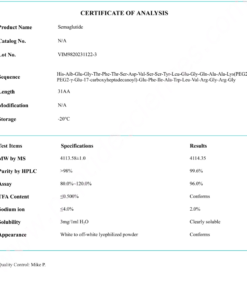
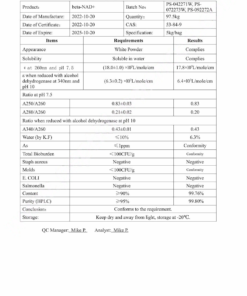
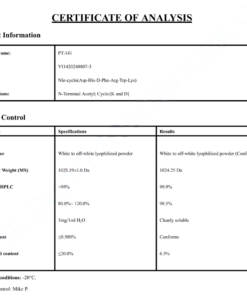
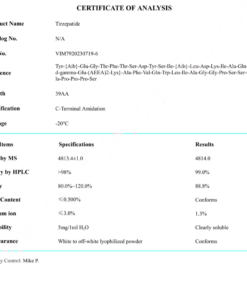
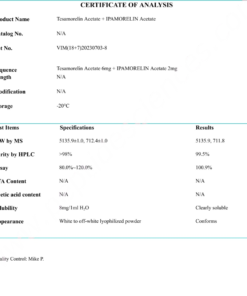
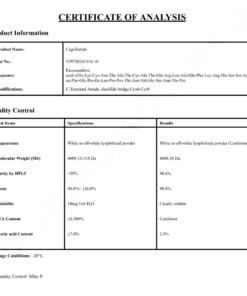
COA Semaglutide (GLP-1 Analogue) Certificate
Semaglutide (GLP-1 Analogue) — Clinically Proven Medication for Type 2 Diabetes & Weight Management
Semaglutide is a next-generation GLP-1 receptor agonist (glucagon-like peptide-1 analogue) indicated for the treatment of type 2 diabetes mellitus and chronic weight management in adults. Administered as a once-weekly subcutaneous injection or as a daily oral tablet, semaglutide delivers superior glycaemic control, meaningful weight reduction, and proven cardiovascular protection — making it one of the most prescribed and trusted medications in modern endocrinology.
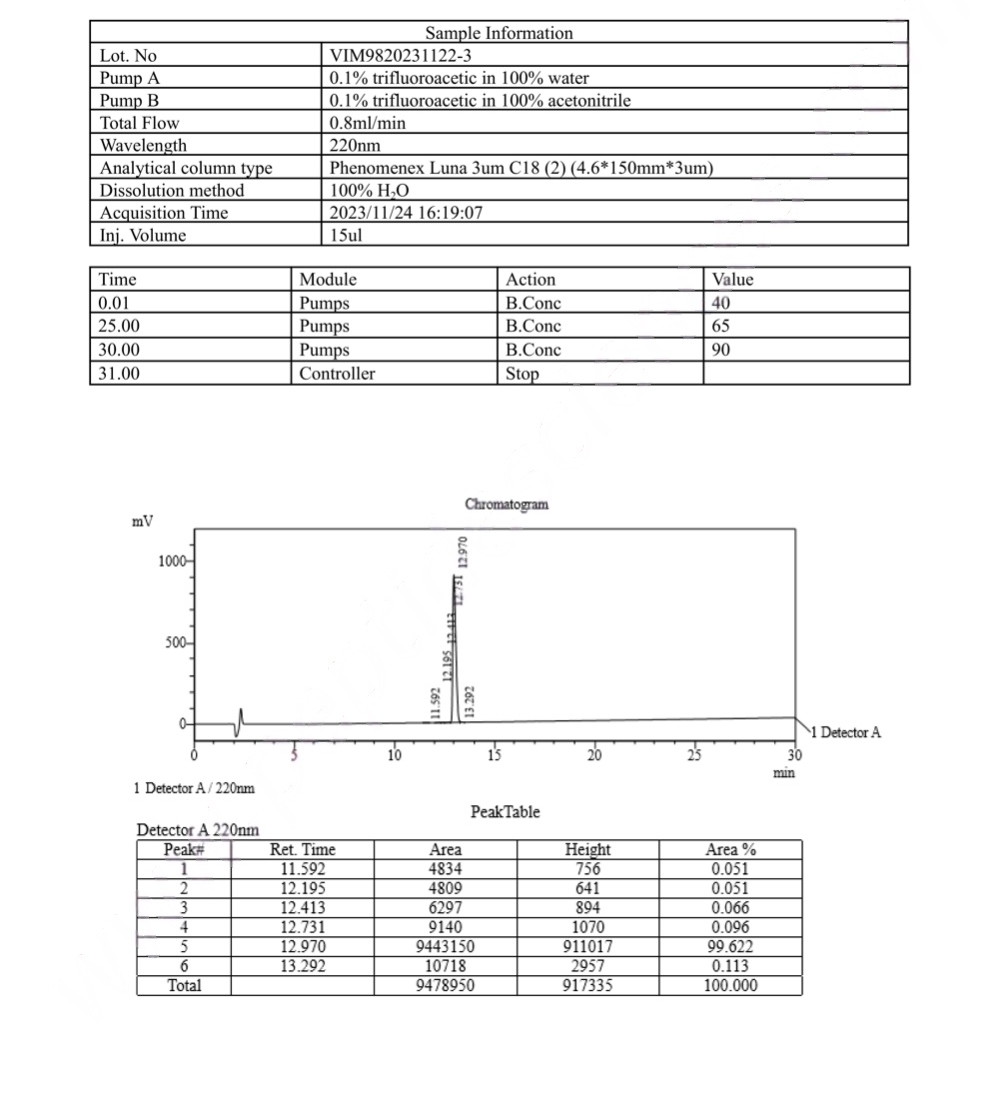
HPLC Semaglutide (GLP-1 Analogue) Certificate
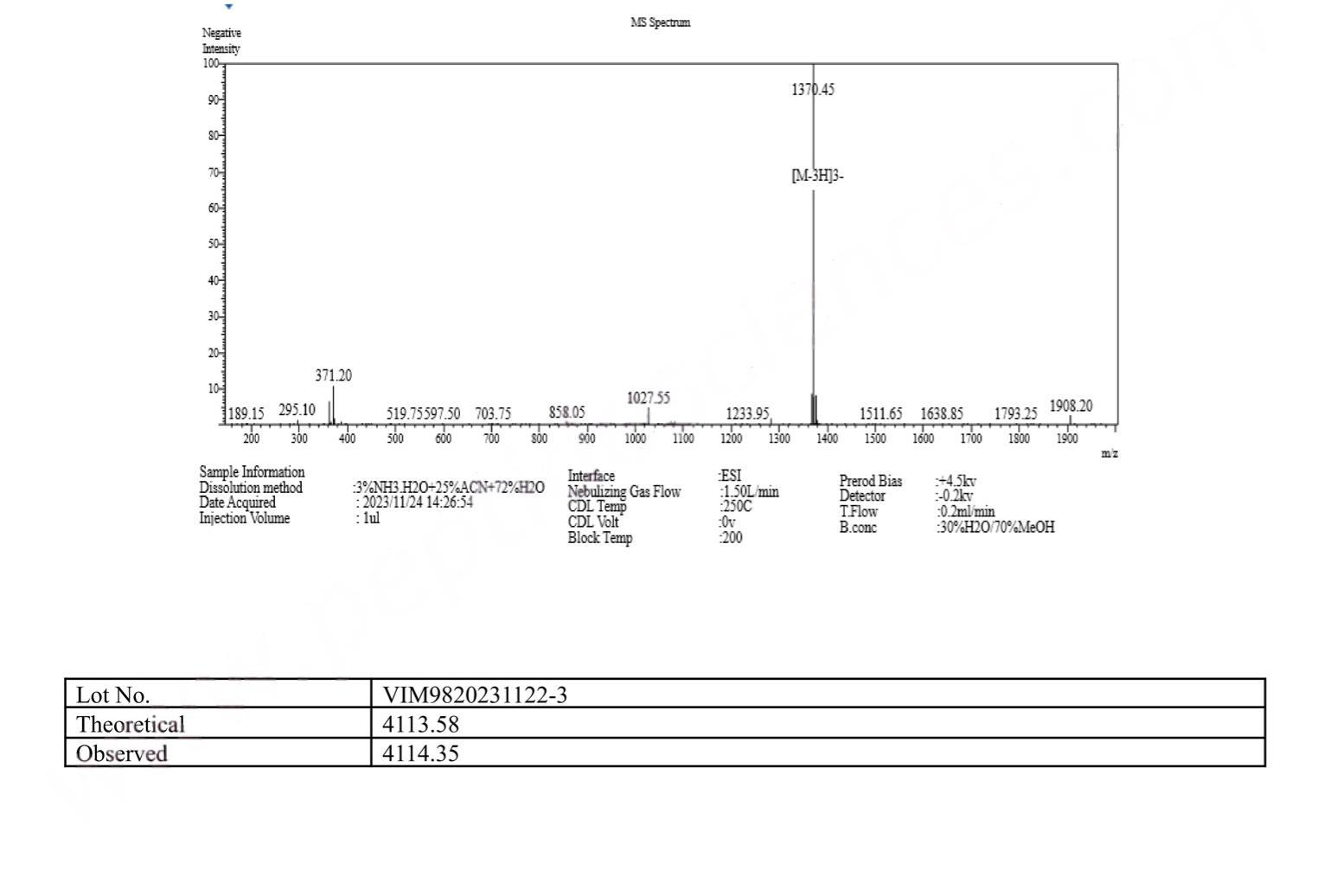
MS Semaglutide (GLP-1 Analogue) certificate
What Is Semaglutide? Understanding the GLP-1 Analogue Mechanism
Semaglutide is a synthetic analogue of the endogenous incretin hormone GLP-1, structurally modified with a C-18 fatty acid chain to extend its half-life to approximately 7 days. This modification allows for convenient once-weekly dosing while maintaining sustained pharmacological activity.
How semaglutide works in the body:
- Stimulates glucose-dependent insulin secretion from pancreatic beta cells, reducing the risk of hypoglycaemia.
- Suppresses inappropriate glucagon release, preventing excess hepatic glucose output.
- Slows gastric emptying, leading to lower post-meal blood glucose spikes.
- Acts on hypothalamic appetite centres to promote satiety and reduce caloric intake.
- Supports beta-cell preservation and function over time.
Approved Indications: What Is Semaglutide Used For?
Semaglutide is approved by the FDA and EMA for:
- Type 2 Diabetes Mellitus (T2DM): to improve glycaemic control as an adjunct to diet and exercise.
- Chronic Weight Management (Obesity / Overweight): in adults with a BMI ≥ 30 kg/m², or ≥ 27 kg/m² with at least one weight-related comorbidity.
- Cardiovascular Risk Reduction: to lower the risk of major adverse cardiovascular events (MACE) in adults with T2DM and established cardiovascular disease.
Key Clinical Benefits of Semaglutide GLP-1 Therapy
Clinical trials including the SUSTAIN and STEP programmes have confirmed semaglutide’s broad therapeutic profile:
Superior Blood Sugar Control
Semaglutide consistently reduces HbA1c by up to 1.8% from baseline — one of the most significant reductions among GLP-1 receptor agonists available. Patients experience fewer blood glucose fluctuations throughout the day, supporting long-term diabetes management and reducing the risk of diabetic complications.
Clinically Significant Weight Loss
In the landmark STEP 1 trial, participants receiving semaglutide 2.4 mg achieved an average body weight reduction of 14.9% over 68 weeks — far exceeding placebo. For individuals with obesity or weight-related conditions, semaglutide represents a transformative option when combined with lifestyle intervention.
Cardiovascular Protection
The SUSTAIN-6 cardiovascular outcomes trial demonstrated a 26% reduction in major adverse cardiovascular events (MACE) with semaglutide versus placebo. This benefit positions semaglutide as a preferred agent for patients with T2DM at high cardiovascular risk.
Semaglutide Dosage, Forms & Administration Guide
Injectable Semaglutide (Subcutaneous)
- Starting dose: 0.25 mg subcutaneously once weekly for 4 weeks.
- Maintenance dose: Escalated to 0.5 mg, then 1 mg, and up to 2 mg weekly depending on clinical response and tolerability.
- Administered in the abdomen, thigh, or upper arm.
- Does not require refrigeration after first use (check product-specific storage instructions).
Oral Semaglutide (Tablet)
- Starting dose: 3 mg once daily for 30 days.
- Escalated to 7 mg, then 14 mg once daily as required.
- Must be taken on an empty stomach with no more than 120 ml of plain water, at least 30 minutes before the first food or drink of the day.
- Do not split, crush, or chew the tablet.
Semaglutide Side Effects: What to Expect
Semaglutide is generally well-tolerated. The majority of side effects are mild-to-moderate and gastrointestinal in nature, occurring most often during dose escalation:
Common Side Effects (≥ 5% of patients)
- Nausea
- Vomiting
- Diarrhoea
- Constipation
- Abdominal pain or discomfort
- Decreased appetite
These symptoms typically resolve within the first few weeks as the body adjusts to therapy. Slow dose titration helps minimise discomfort.
Important Safety Warnings
- Pancreatitis: Discontinue if acute pancreatitis is suspected.
- Thyroid C-cell tumours: Contraindicated in patients with a personal or family history of medullary thyroid carcinoma (MTC) or MEN2.
- Diabetic Retinopathy: Rapid improvements in glycaemic control may worsen diabetic retinopathy in predisposed patients.
- Renal Impairment: Monitor renal function during therapy initiation.
Who Is Semaglutide Right For? Patient Eligibility
Ideal Candidates for Semaglutide Therapy
- Adults with type 2 diabetes not adequately controlled on metformin or other oral agents.
- Individuals with obesity (BMI ≥ 30) or overweight (BMI ≥ 27) with comorbidities such as hypertension or dyslipidaemia.
- Patients with established cardiovascular disease seeking additional risk reduction.
- Those seeking a once-weekly injection for convenience and adherence.
Contraindications: When Semaglutide Should Not Be Used
- Personal or family history of medullary thyroid carcinoma (MTC).
- Multiple Endocrine Neoplasia syndrome type 2 (MEN2).
- Type 1 diabetes mellitus or diabetic ketoacidosis.
- Known hypersensitivity to semaglutide or any excipient.
- Pregnancy or planned pregnancy (discontinue at least 2 months before conception).
Why Choose Semaglutide Over Other GLP-1 Analogues?
When compared to other GLP-1 receptor agonists such as liraglutide, dulaglutide, or exenatide, semaglutide consistently demonstrates:
- Greater HbA1c reductions across head-to-head clinical trials.
- Superior weight loss outcomes, particularly at the 2.4 mg weekly dose.
- Once-weekly subcutaneous dosing for maximum patient convenience.
- Strong cardiovascular outcomes data with MACE reduction.
- Availability in both injectable and oral formulations — the world’s first oral GLP-1 agonist.
Storage, Handling & Shelf Life of Semaglutide
Injectable Pen Storage
- Unopened pens: Store in a refrigerator at 2°C–8°C.
- Once in use: Store at room temperature (below 30°C) for up to 6 weeks.
- Keep away from heat, sunlight, and freezing temperatures.
- Do not use if the solution appears cloudy, discoloured, or contains particles.
Oral Tablet Storage
- Store below 30°C in the original blister packaging.
- Keep out of reach of children.
- Do not use past the stated expiry date.
| Quantity | 5mg * 10 vials, 10mg * 10 vials, 15mg * 10 vials, 20mg * 10 vials, 30mg * 10 vials, 60mg * 10 vials, 100mg * 10 vials, 4mg Pens, 8mg Pens |
|---|
Be the first to review “Semaglutide” Cancel reply
Related products
Popular products
Peptide
Popular products
Popular products
Popular products
Popular products
Popular products
Popular products

















Reviews
There are no reviews yet.